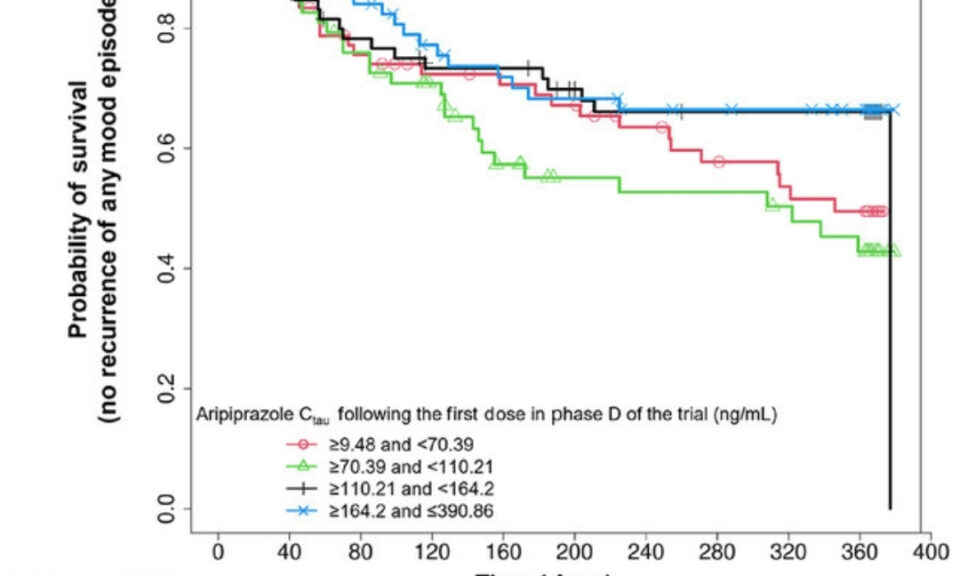
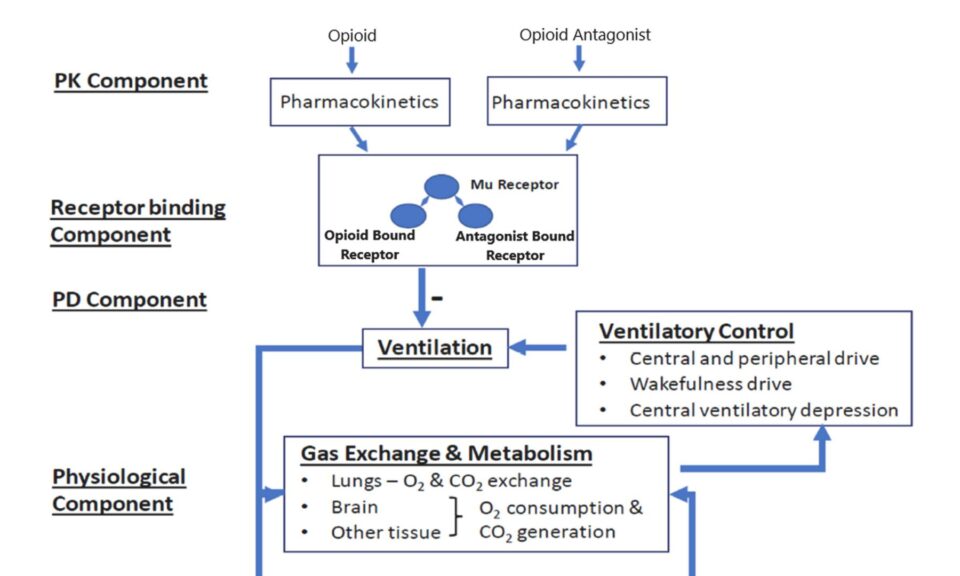
Stefan Roepcke, Ph.D.
Senior Director, Pharmacometrics
Clinical Pharmacology & Pharmacometrics Solutions
About Stefan
Stefan has solid pharmacometrics and biostatistics skills, with 14 years of experience in clinical and preclinical drug development in the pharmaceutical industry.
Through his work, he supports:
- clinical trial planning and dose justification
- clinical data mining for repurposing
- quantitative support for well-grounded management decisions
- communication of model-based results to decision bodies and authorities
He acquired specific knowledge in the therapeutic areas of oncology, immunology & inflammation, pain, diabetes, CNS, and respiratory and gastrointestinal, developing small molecules and biologics during his career. Regulatory approvals and scientific publications prove the quality of his contributions. He has a track record of successful collaborations and established links to clients and partners. He has published numerous scientific papers in peer-reviewed journals, has presented at national and international conferences, and teaches courses in pharmacometrics and biostatistics.