This second part of the REST API series demonstrates how different client applications—from Spotfire to Python scripts—interact with the ADMET Predictor REST API. Watch real-time examples of HTTP requests and responses, including property predictions, image generation, and pharmacokinetic simulations.

ADMET Predictor® Tutorial Series: Rest API Part 1
This first part of a two-video series explains how to set up the ADMET Predictor REST API server. Follow the step-by-step process for installing the Windows service, configuring license server paths, and managing support folders for seamless property predictions in third-party tools.

ADMET Predictor® Tutorial Series: Transporter Models
This tutorial by Simulations Plus focuses on the Transporters Module in ADMET Predictor®. Understand the role of transporters in drug exposure and toxicity, and learn how to use the 24 built-in substrate and inhibitor models to screen compound libraries.

ADMET Predictor® Tutorial Series: Toxicity Predictions
This tutorial covers the comprehensive Toxicity Module in ADMET Predictor®. Learn about hERG channel inhibition, Ames mutagenicity strains, and how these individual models contribute to the overall Tox Risk score.

ADMET Predictor® Tutorial Series: Cheminformatics Part 2
In this second part of the Cheminformatics series, Simulations Plus demonstrates how to utilize pre-built CQF (Compound Query Files) for drug-like filtering and toxicophore detection. Learn to customize these files using SMARTS syntax to create personalized structural filters, such as Michael acceptor or PAINS filters.

ADMET Predictor® Tutorial Series: Cheminformatics Part 1
In this first part of the Cheminformatics series, we explore core tools for chemical space exploration. Learn how to perform explicit substructure searches and advanced Markush queries to categorize large datasets by molecular features.

ADMET Predictor® Tutorial Series: AIDD Module
This tutorial by Simulations Plus introduces the Artificial Intelligence Drug Design (AIDD) module. Discover how to optimize lead compounds for potency, synthetic feasibility, and ADMET properties using multi-objective Pareto optimization.

ADMET Predictor® Tutorial Series: ADMET Modeler
This comprehensive tutorial by Simulations Plus demonstrates the workflow for building custom machine learning models using ADMET Modeler™. Learn how to prepare data, select descriptors, set up artificial neural network (ANN) ensembles, and validate models with external test sets

ADMET Predictor® Tutorial Series: HTPK Part 3
In the final part of the HTPK series, Simulations Plus demonstrates how to generate Concentration-Plasma (CP) time profiles for multiple compounds and doses. This tutorial also covers Dose Estimation to meet target plasma concentrations and explores advanced physiological and formulation settings.

ADMET Predictor® Tutorial Series: HTPK Part 2
In part two of the HTPK series, we explore the Parameter Sensitivity Analysis (PSA) tool to determine whether fraction absorbed is limited by solubility or permeability. The video also demonstrates how to increase simulation accuracy by integrating experimental data alongside in silico predictions

Population Pharmacokinetic, Exposure-Response, and Time-To-Event Analyses of Probenecid in Symptomatic, Non-Hospitalized Patients with COVID-19
Probenecid, an oral uricosuric agent, is being developed as a broad-spectrum antiviral and was evaluated for potential suppressive effects of SARS-Cov-2 replication in a Phase 2 study in patients with symptomatic, mild-to-moderate COVID-19

Comparison of Population Pharmacokinetic Platforms, Monolix and Phoenix for Cephalexin in Infants
Cephalexin is a commonly used antibiotic for pediatric populations, however, data to guide effective oral dosing in young infants are limited.
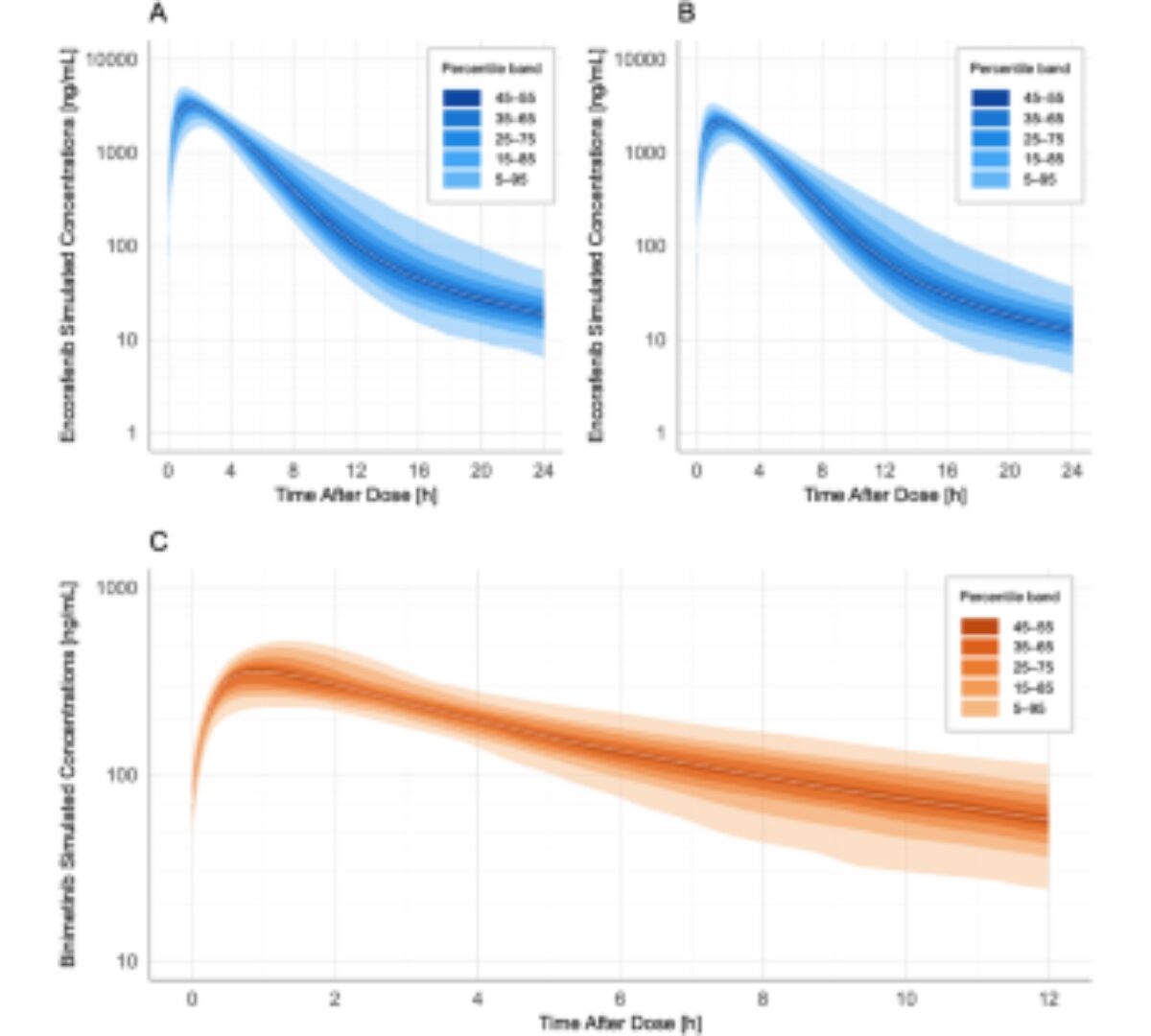
Population Pharmacokinetics of Encorafenib and Binimetinib in Real-World Patients with BRAFV600E/K-Mutant Metastatic Melanoma
Encorafenib and binimetinib pharmacokinetic (PK) studies in real-world cancer patients remain scarce.
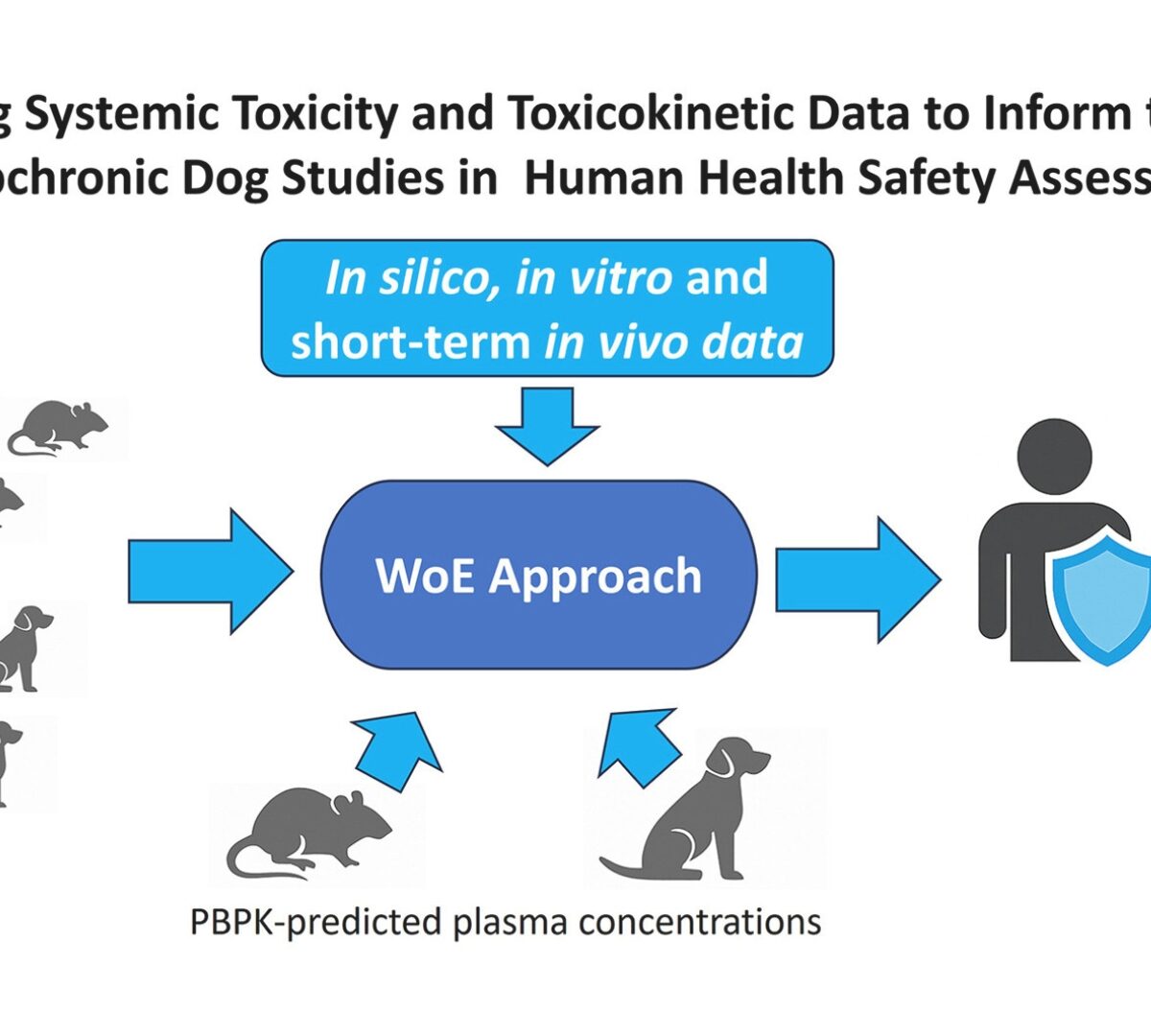
Integrating Systemic Toxicity and Toxicokinetic Data to Inform the Need for Subchronic Dog Studies in Human Health Safety Assessments of Agrochemicals
Regulatory testing for agrochemicals has traditionally included a 90-day toxicity study in a non-rodent species, usually the dog.

A Phase 1 Evaluation of Inhaled Oxytocin: Physiologically-Based Pharmacokinetic Model Informed Dosing of a Novelheat-Stable Oxytocin Delivery System
To develop and validate a physiologically-based pharmacokinetic (PBPK) modelenabling inhaled oxytocin dose selection for clinical evaluation.

ADMET Predictor® Tutorial Series: HTPK Part 1
In this first part of the HTPK series, we demonstrate how to run high-throughput pharmacokinetic (HTPK) simulations directly from chemical structures. Learn to calculate fraction absorbed (%FA), fraction bioavailable (%FB), and key PK parameters like Cmax, Tmax, and clearance across multiple doses.

ADMET Predictor® Tutorial Series: MedChem Studio™ Part 2
In the second part of the MedChem Studio™ series, we demonstrate how to evaluate compound subsets and optimize virtual libraries. This tutorial covers distribution analysis to compare virtual vs. exemplified compounds and introduces matched molecular pair (MMP) analysis for property cliff detection.

ADMET Predictor® Tutorial Series: MedChem Studio™ Part 1
In this first installment of the MedChem Studio™ series, we demonstrate powerful clustering and group analysis tools. Learn how to organize large data sets into chemotypes, align scaffolds for visual comparison, and perform R-group decomposition to identify SAR trends

ADMET Predictor® Tutorial Series: Predicting Metabolites
In this video, we explore advanced tools for predicting drug metabolites across major enzyme classes, including Cytochrome P450 (SIP), Aldehyde Oxidase (AOX), and UGTs. You will learn how to identify atomic sites of metabolism, interpret scoring systems, and visualize structural transformations within MedChem Designer™.

ADMET Predictor® Tutorial Series: Model Editor
In this video, we provide a detailed overview of the Model Editor, explaining how to manage prediction modules and interpret applicability domains. Key topics include descriptor sensitivity analysis (DSA), structure sensitivity analysis (SSA), and regression uncertainty.
