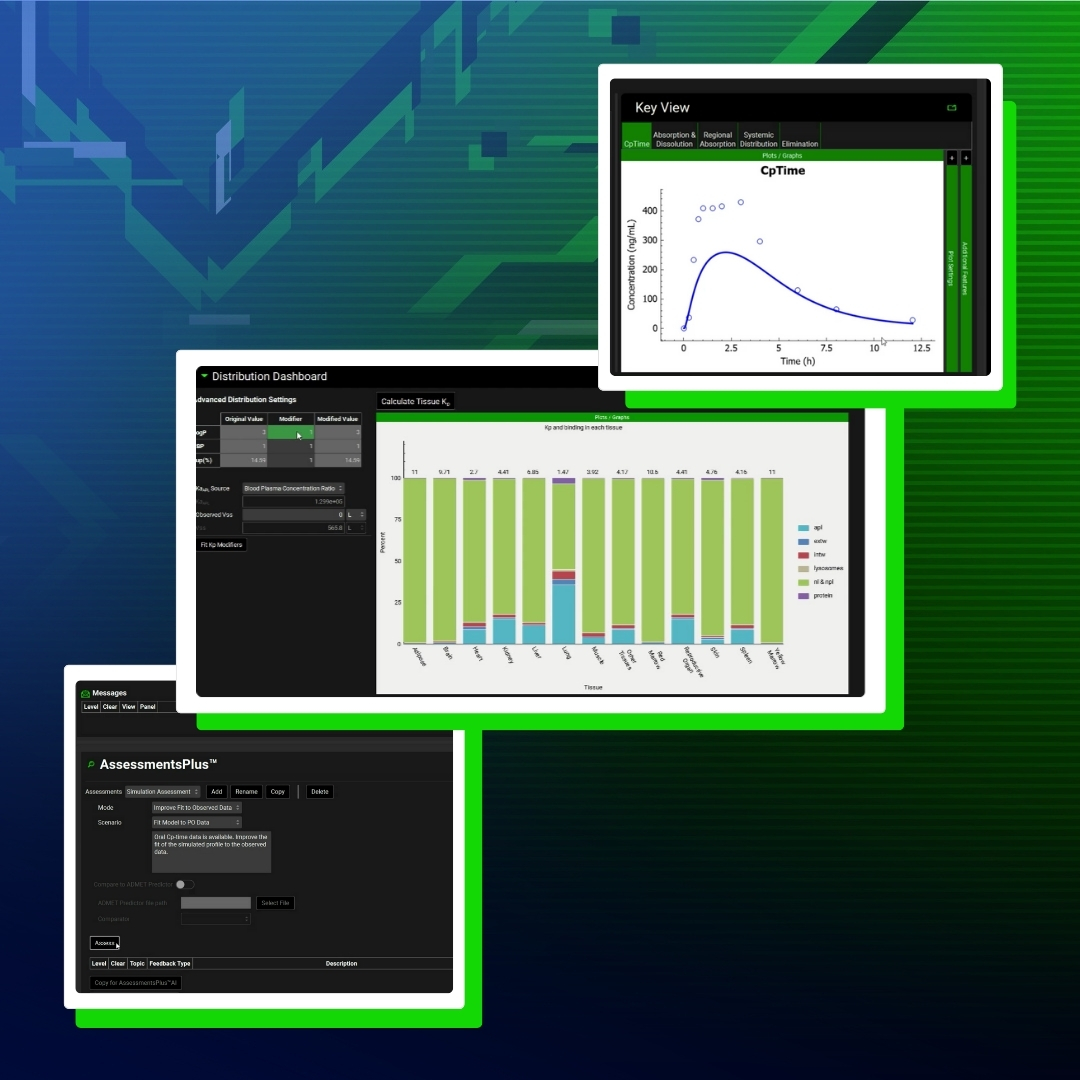
Mechanistic biopharmaceutics modeling: trusted, flexible, and built to scale.

DILIsym® 11 Product Brochure
Quantitative Systems Toxicology (QST) Software for Predicting and Explaining Drug-Induced Liver Injury (DILI)
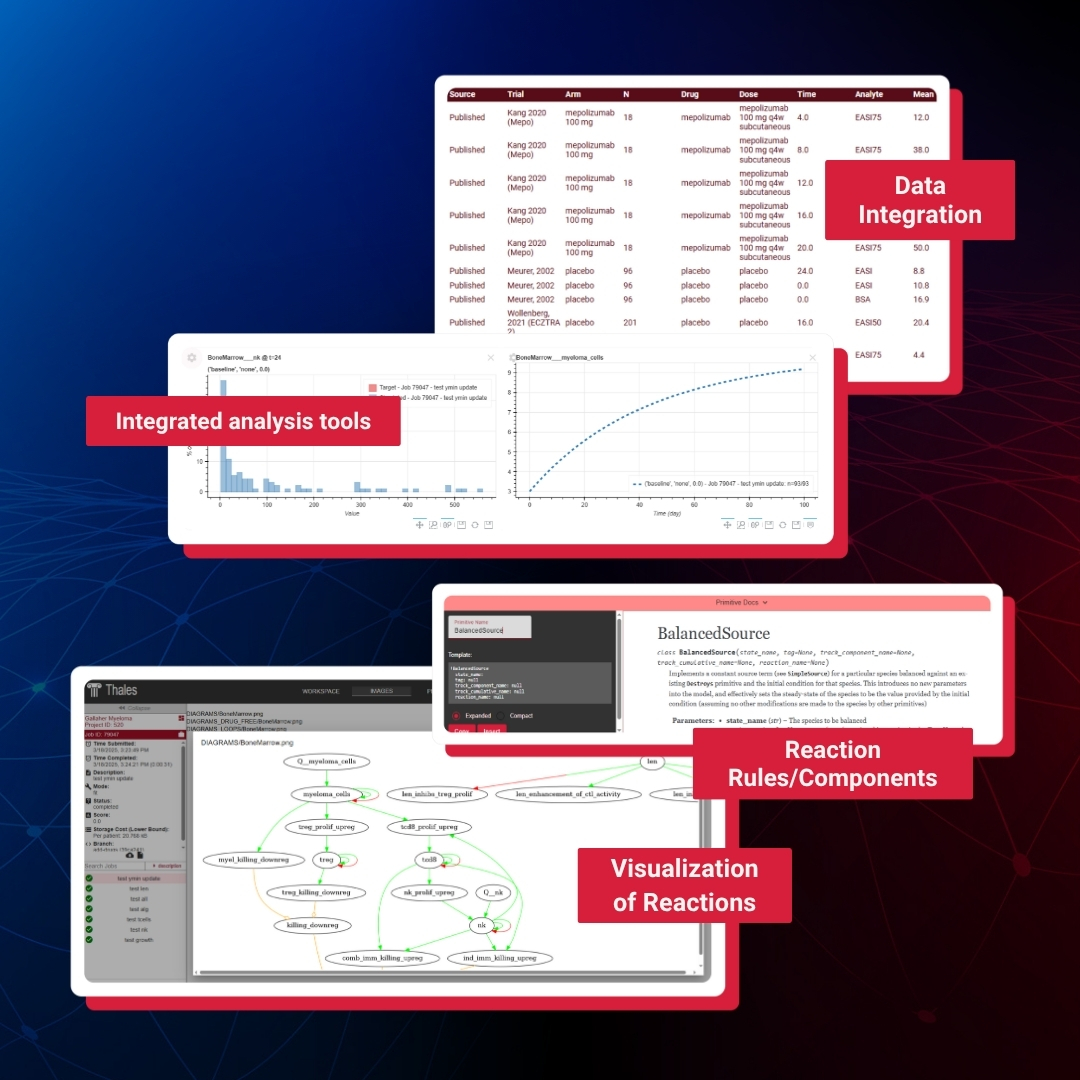
Thales™ Product Brochure
Accelerate your model development
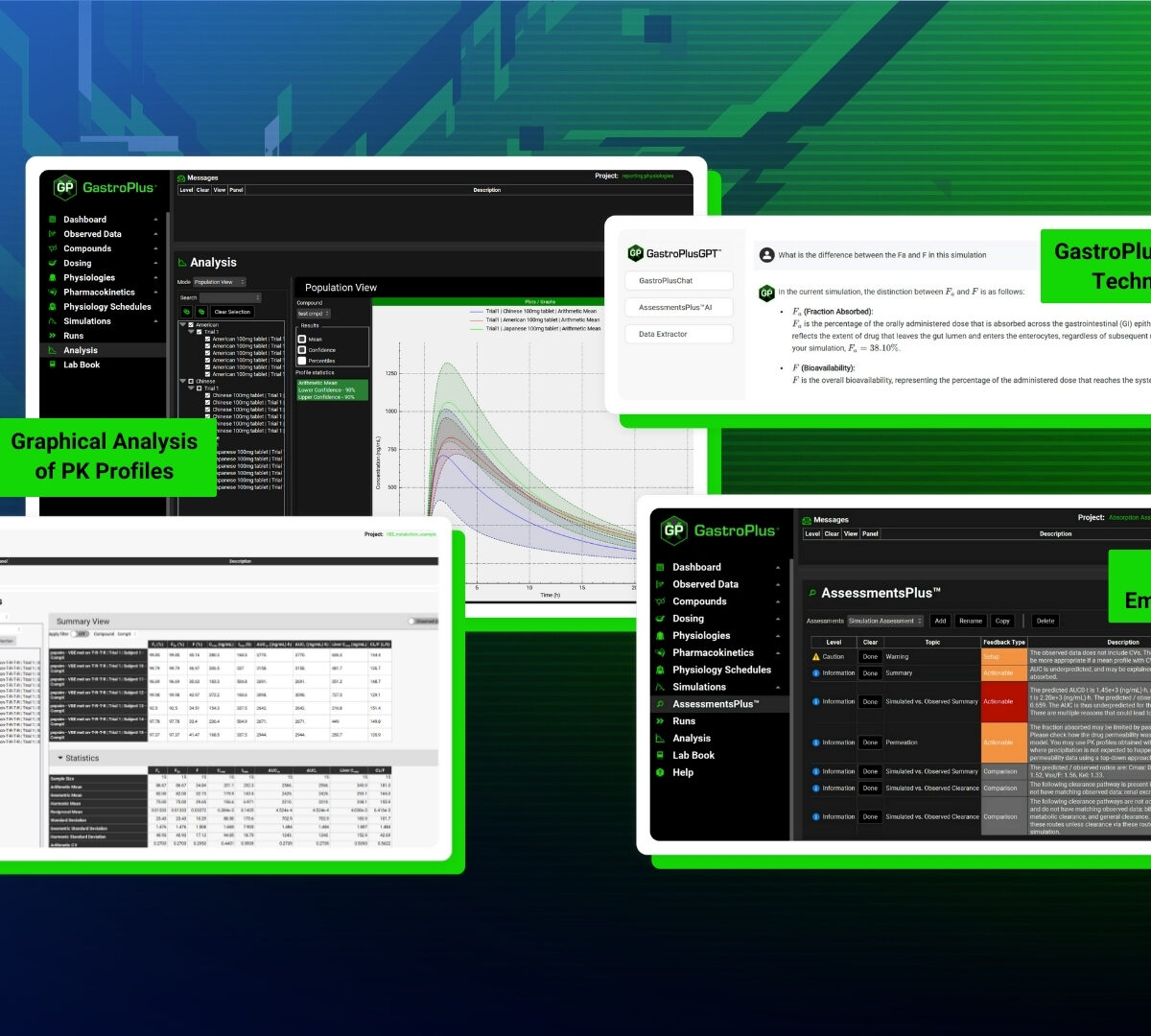
GastroPlus®X.2 Product Brochure
GastroPlus X.2 is a mechanistically based simulation software package that simulates intravenous and oral absorption, pharmacokinetics, drug-drug interactions and pharmacodynamics in humans and animals.

ADMET Predictor® 13 Product Brochure
Proven algorithms. Premium data. Predictions you can rely on.

AP Transporters Module Flyer
Transporters Module enables data-driven decision-making by leveraging AI/machine learning (ML) models trained on premium experimental datasets.

Liver Safety+ Flyer
The only platform that allows you to predict liver safety during discovery

Advancing Scientific Development with Collaborations
15+ FDA grants and $5M+ in funding & new feature developments since 2014

GP AP Module Flyer
The ADMET Predictor Module uses enhanced pk models developed with greater accuracy. It uses the same QSPR models as our best-inclass ADMET Predictor standalone software.

GP DDI Module Flyer
The DDI Module in GastroPlus allows you to predict mechanistic and static drug-drug interactions (DDIs) among unlimited drugs and metabolites.
GP PKPlus™ Module Flyer
The fitted parameters include PK properties, first orderabsorption rate, bioavailability and absorption lag time.
RENAsym Flyer
RENAsym Predict and Prevent Drug-Induced Kidney Injury
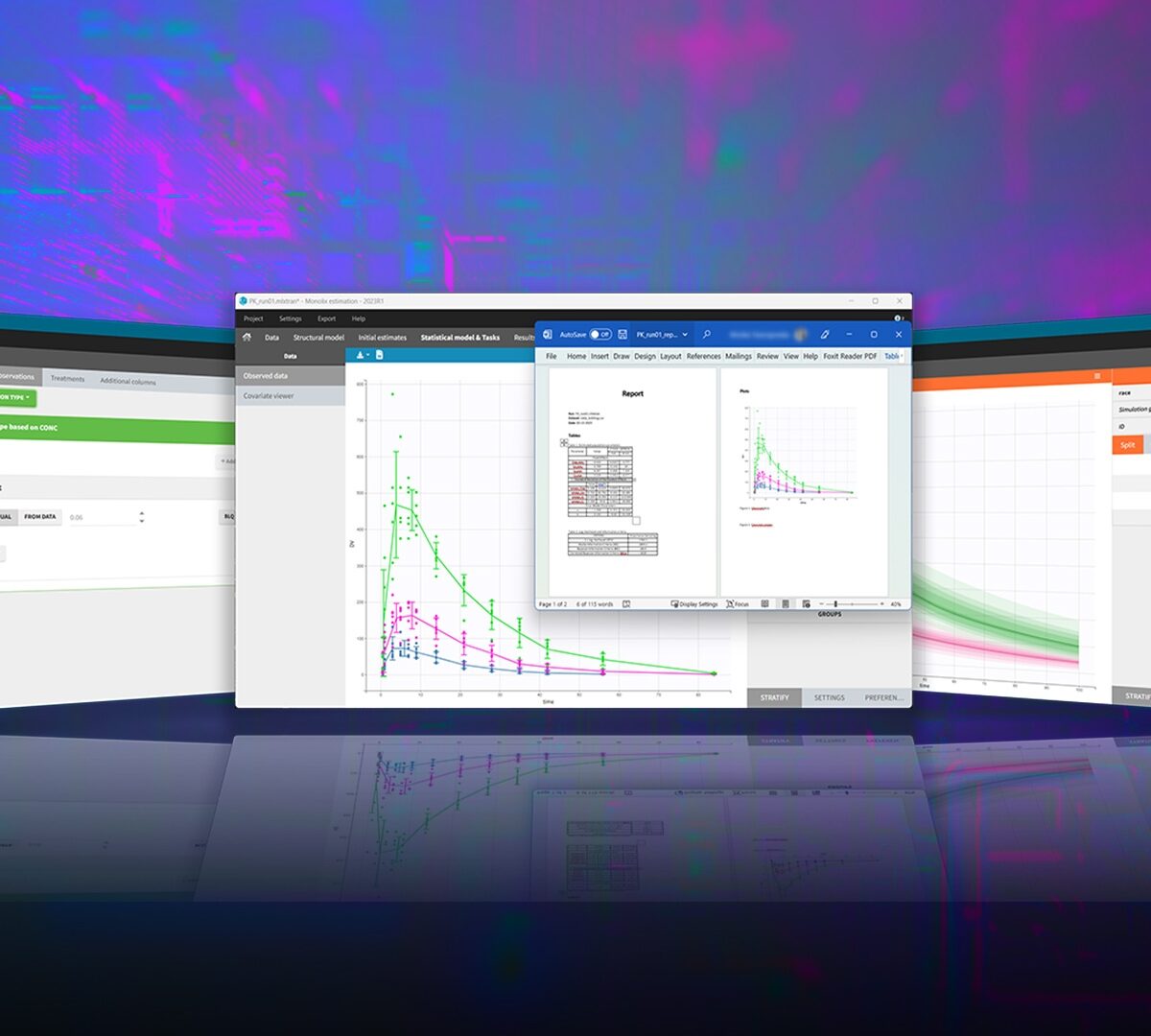
MonolixSuite™ v2024 Product Brochure
A fast easy-to-use and powerful suite of applications for pharmacometrics analysis, modeling and simulation.

EvolvePK+ Comparison Flyer
Only one platform can integrate GastroPlus PBPK outcomes during early design and lead optimization.

The Power of Simulation Training
Experience in Clinical Trial Training
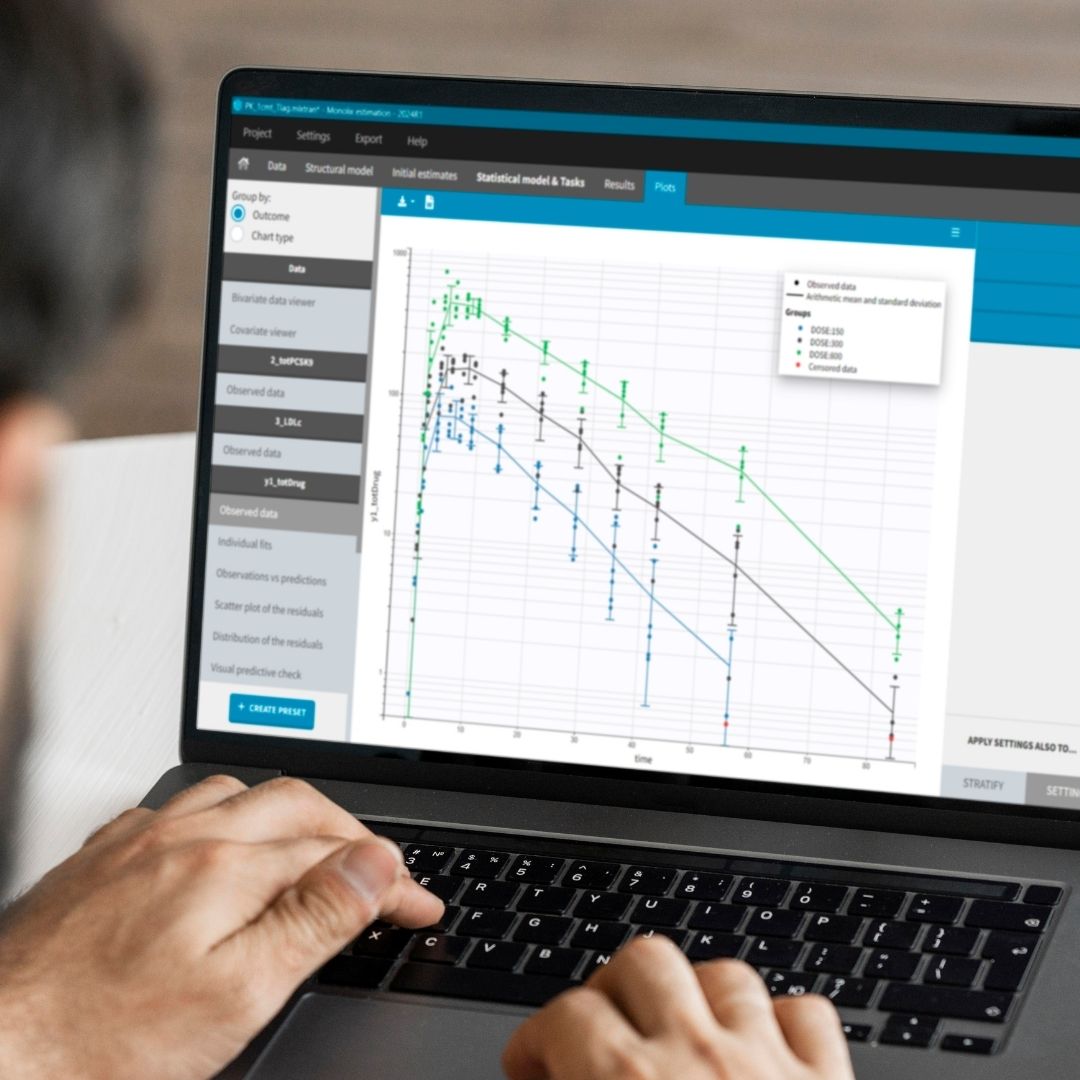
Monolix Comparison Flyer
Your software should make it easier to conduct non-compartmental, bioequivalence and compartmental analysis and use that data to make the right decisions for your program.

Regulatory Strategies Flyer
Get to Market Faster with Robust Regulatory Strategies

Clinical Pharmacology & Pharmacometrics Software and Services Flyer
Supporting Dose Selection and Justification
Cheminformatics Software & Services Flyer
Understand Your Compound