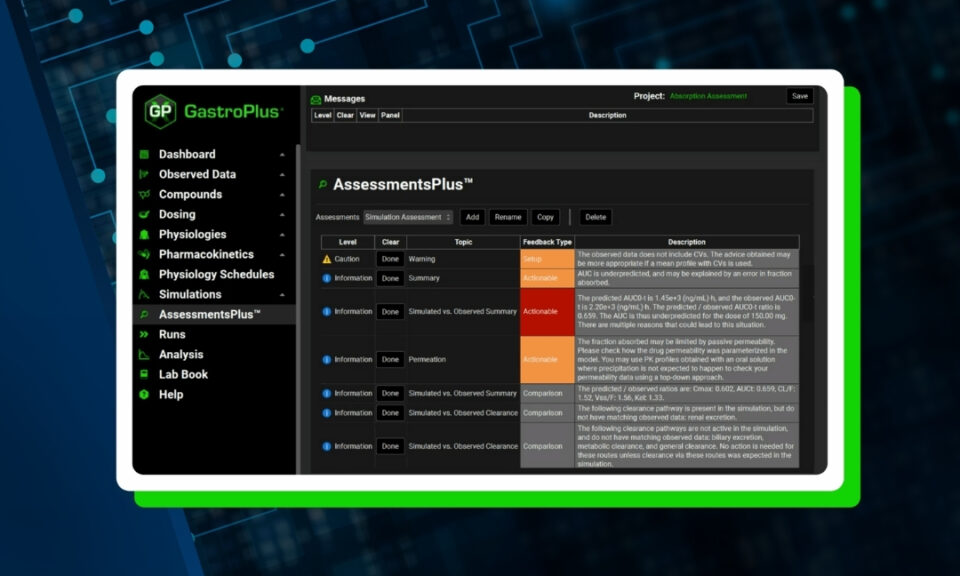
Simulations Plus offers PBPK modeling and simulation consulting services using our GastroPlus® software – the pharmaceutical industry’s most sophisticated platform for the prediction of drug absorption and disposition in human and animal species. GastroPlus, with its #1-ranked Advanced Compartmental Absorption and Transit (ACAT™) and PBPK models, has features and capabilities to support Model Based Drug Development in all phases of drug discovery, translational research, and clinical development.
If your organization does not have the time or resources to use our software directly, or if you need a fast turnaround on a particular project, our comprehensive consulting service is the solution. Our interdisciplinary team of experts can review your data, work with you to identify the issues of concern, and apply our technology and expertise to offer unique insights that will guide your projects towards an optimal strategy and outcome.
The combination of PBPK modeling techniques with our population PK/PD modeling and simulation expertise allows us to offer a variety of high-value, end-to-end solutions.