Physiologically based pharmacokinetic modelling is routinely used in the pharmaceutical industry and has an impact on drug labels.

Pulmonary Physiological-Based Pharmacokinetic (PBPK) Modeling for Clinical Systemic and Regional PK Prediction of Inhaled Nintedanib Drug Products
Nintedanib (Nint), a tyrosine kinase inhibitor, is approved by the FDA for the treatment of idiopathic pulmonary fibrosis (IPF) by administration as an oral tablet.

Mechanistic PBPK Modeling Identifies Oral Microenvironment Determinants of Buccal Midazolam Exposure
Buccal drug delivery relies on prolonged mucosal contact for absorption, sensitive to interindividual chemical and physical variability within the oral microenvironment (Hua, 2019; Shipp et al., 2022).

Development of a Physiologically Based Pharmacokinetic (PBPK) Simulation Model for Nicotine
Tobacco is a major cause of chronic diseases such as lung cancer, cardiovascular disease, and chronic obstructive pulmonary disease worldwide.

Physiologically Based Pharmacokinetic Modeling to Predict Human Pharmacokinetics of a Novel Mithramycin Analog for Ewing Sarcoma
To develop and verify a physiologically based pharmacokinetic (PBPK) modeling strategy for mithramycin (MTM) and its analog, MTMSA-Trp, with the aim of projecting first-in-human plasma pharmacokinetics and supporting the translational development of MTMSA-Trp for Ewing sarcoma treatment.

Reproductive Toxicity of Fructus Psoraleae in Zebrafish: Material Basis and Implications for Clinical Safety Dosing
Fructus psoraleae (FP), a classical TCM with a medicinal history spanning several millennia, is esteemed for its therapeutic properties in regulating Qi, alleviating asthma, tonifying kidneys and reinforcing Yang.

Physiologically Based Pharmacokinetic Modeling and Dose Adjustment of Imipenem in Pediatric Patients with Renal Impairment Chen Feng Chen Feng 1 P Peng Xiao 1,2
To establish a physiologically based pharmacokinetic (PBPK) model of imipenem, predict its exposure in pediatric patients with different renal function, and optimize the dosing regimen.

A Drug–Microbiome–Drug Interaction Impacts Co-Prescribed Medications for Parkinson’s Disease
Simultaneous prescription of multiple drugs is widespread in medicine. Although the gut microbiome is implicated in drug responses, its role in mediating drug–drug interactions is unexplored

Nanoformulation Enabling Repurposing of Niclosamide for the Treatment of Pancreatic Cancer: Rapidly Soluble Oral Formulation for Enhanced Bioavailability
Pancreatic ductal adenocarcinoma (PDAC) is relatively uncommon yet remains one of the most lethal cancers worldwide.

Nonlinear Mixed-Effects Modeling to Characterize the Pharmacokinetics of a Novel Mithramycin Analogue for Ewing Sarcoma in Mice
To develop a pharmacokinetic model for a novel mithramycin analogue, MTMSA-Trp, in mice and characterize dose-dependent disposition to support future pharmacokinetic-pharmacodynamic (PK/PD) and exposure-e cacy analyses.

Piperazine-Thiourea Hybrids as Novel Antiplatelet Agents Targeting COX-1: Synthesis, in Vitro, and in Silico Evaluation
Cardiovascular diseases (CVDs) remain the leading cause of mortality worldwide, reinforcing the need for safer and more effective antiplatelet therapies.

Pharmacokinetic Evaluation of Etoricoxib 120mg Tablets in Healthy Human Pakistani Volunteers: In-Vivo In-Silico Bridging for Bioequivalence
Etoricoxib is a selective cyclooxygenase-2 inhibitor widely used for the treatment of pain and inflammatory conditions.

In Silico Designing of Palbociclib Loaded Plga Long-Acting Intramuscular Injection for Palliative Therapy of HR+/HER2− Metastatic Breast Cancer
Palbociclib (PBB) is an oral cyclin-dependent kinase 4/6 (CDK4/6) inhibitor approved for the treatment of HR+/HER2− breast cancer.

Comparison of Population Pharmacokinetic Platforms, Monolix and Phoenix for Cephalexin in Infants
Cephalexin is a commonly used antibiotic for pediatric populations, however, data to guide effective oral dosing in young infants are limited.
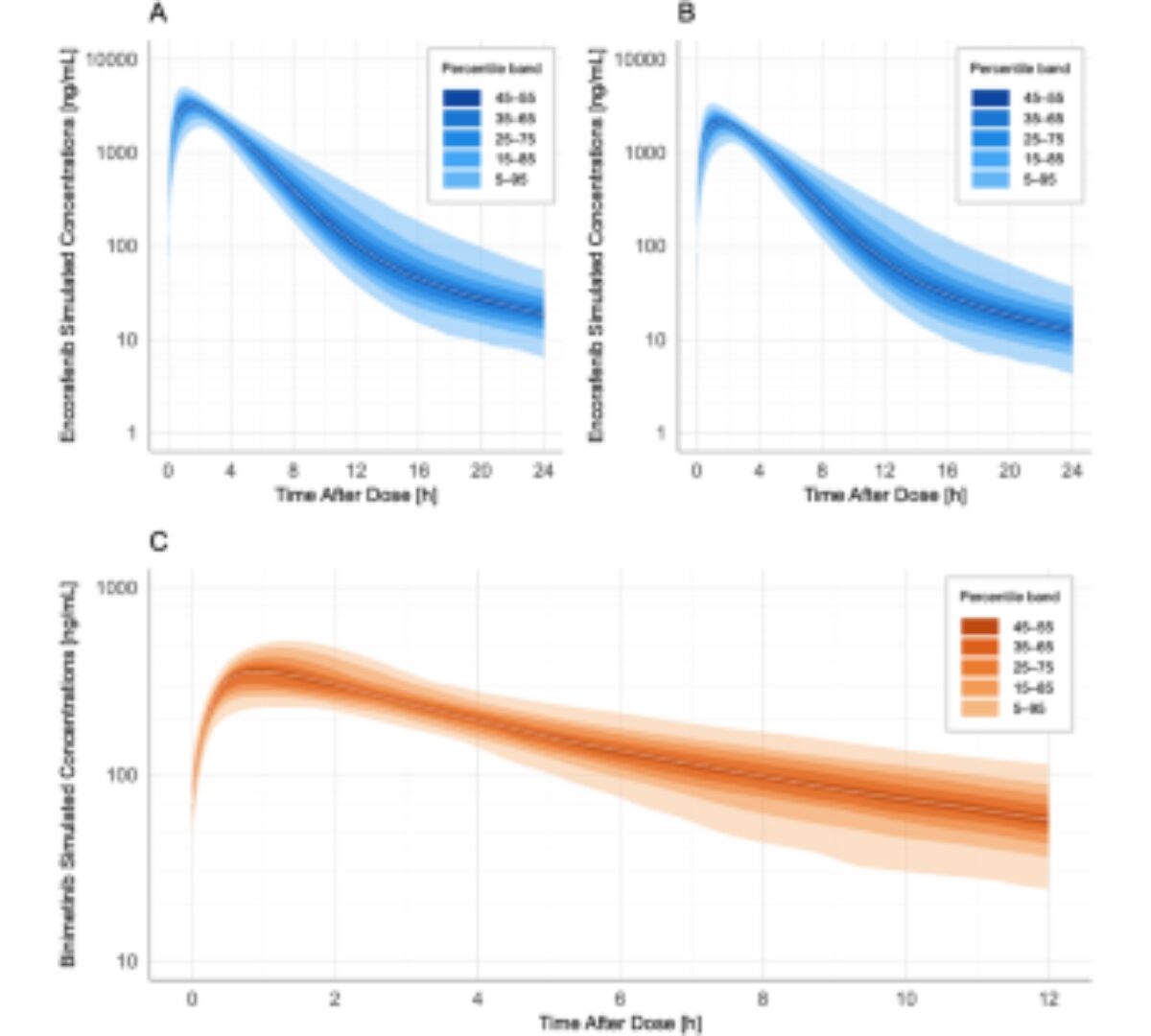
Population Pharmacokinetics of Encorafenib and Binimetinib in Real-World Patients with BRAFV600E/K-Mutant Metastatic Melanoma
Encorafenib and binimetinib pharmacokinetic (PK) studies in real-world cancer patients remain scarce.
Integrating Systemic Toxicity and Toxicokinetic Data to Inform the Need for Subchronic Dog Studies in Human Health Safety Assessments of Agrochemicals
Regulatory testing for agrochemicals has traditionally included a 90-day toxicity study in a non-rodent species, usually the dog.

A Phase 1 Evaluation of Inhaled Oxytocin: Physiologically-Based Pharmacokinetic Model Informed Dosing of a Novelheat-Stable Oxytocin Delivery System
To develop and validate a physiologically-based pharmacokinetic (PBPK) modelenabling inhaled oxytocin dose selection for clinical evaluation.

Prediction of the Lurasidone–Posaconazole Drug–Drug Interaction Using Physiologically Based Pharmacokinetic Modeling
Lurasidone is an atypical antipsychotic drug that metabolized by cytochrome P4503A4 (CYP3A4). Posaconazole is a triazole antifungal agent known to inhibit CYP3A4activity.
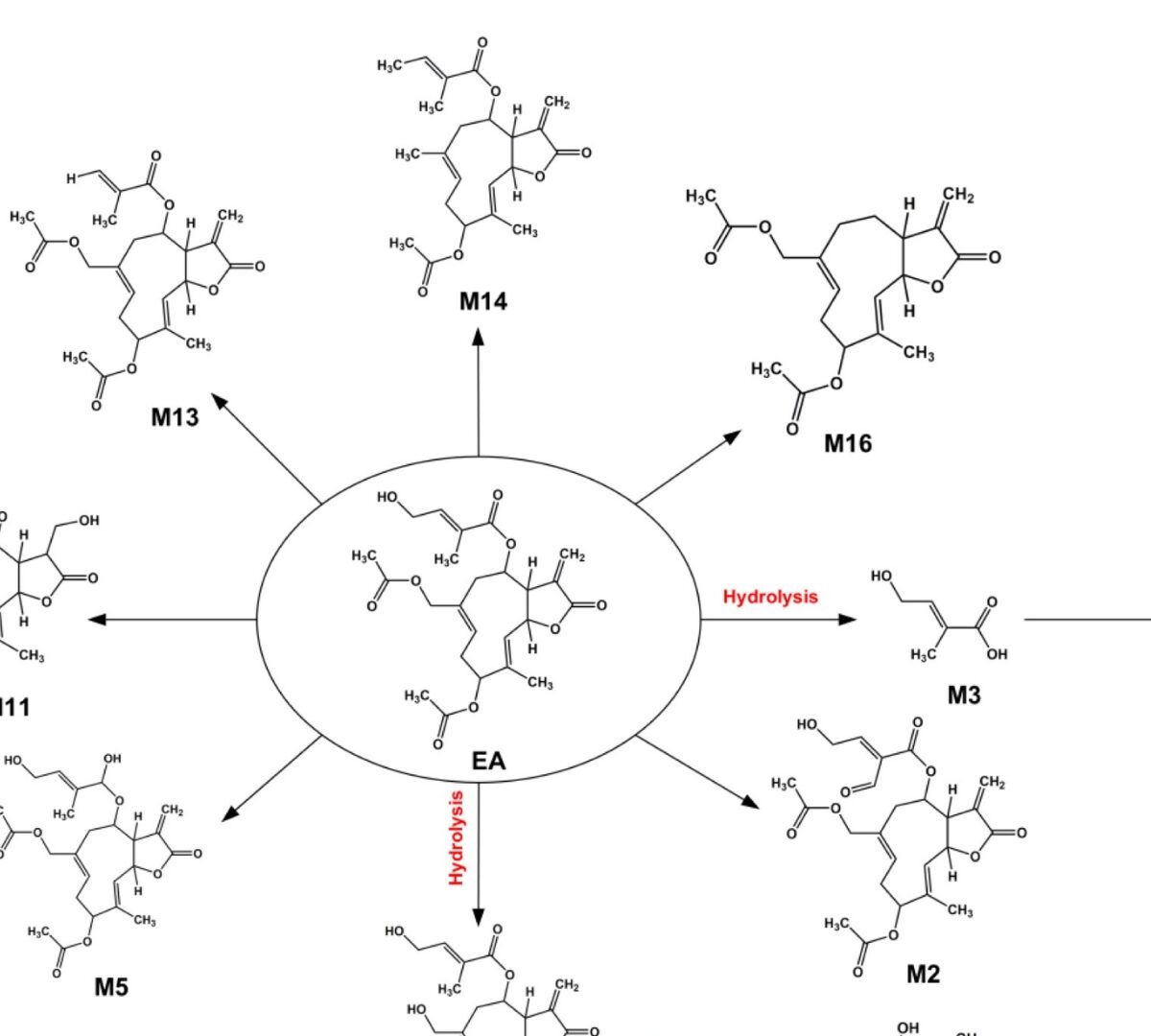
Metabolic Profiling and Detoxification of Eupalinolide A and B in Human Liver Microsomal Systems
Eupalinolide A (EA, Z-configuration) and Eupalinolide B (EB, E-configuration) are cis-trans isomeric sesquiterpenoid monomers isolated from Eupatorium lindleyanum DC. (Asteraceae).

Automated Concentration-QT data preparation, model selection and reporting in R
Since the publication of the ICH E14 guidance in 2015, QT interval prolongation as-sessment can be carried out with a concentration-QTc modeling approach as part of single- or mul-tiple- dose escalation studies, instead of conducting a thorough QT/QTc study.
