The tissue response to long-acting injectables (LAIs) suspension injection may impact the product in vivo performance.

Model-Based Meta-Analysis With MonolixSuite: A Tutorial for Longitudinal Categorical and Continuous Data
Model-based meta-analysis (MBMA) informs key drug development decisions by integrating data, published or unpublished, from multiple studies.

Development of an Improved Dissolution Testing Method as an Alternative to Animal Testing
This study aimed to evaluate the potential of an improved flow-through cell dissolution method as an alternative to animal testing for predicting the in vivo behavior of immediate-release formulations.

Exploring Artificial Intelligence’s Potential to Enhance Conventional Anticancer Drug Development
Cancer affects one in three to four people globally, with over 20 million new cases and 10 million deaths annually, projected to rise to 35 million cases by 2050. Developing effective cancer treatments is crucial, but the drug discovery process is a highly complex and expensive endeavor, with success rates sitting well below 10% for oncologic therapies

Mechanistic Pharmacokinetic Models for Inhaled Drug Development: Pulmonary Physiological and Structural Characteristics and Modeling Approaches
Pulmonary drug delivery via inhalation offers significant clinical benefits including rapid onset of action and targeted drug administration, which minimizes systemic adverse effects.

Simulated Pharmacokinetic Compatibility of Tamoxifen and Estradiol: Insights from a PBPK Model in Hormone-Responsive Breast Cancer
Although traditionally contraindicated, the coadministration of tamoxifen and estradiol may hold clinical relevance in specific contexts, particularly in breast cancer survivors with premature menopause...
![Whole-Body Disposition and Metabolism of [14C]-2,4,4′-Trichlorobiphenyl (PCB28) Following Lung Administration in Rats](https://www.simulations-plus.com/wp-content/themes/simulations-plus/library/dist/img/default_square-large.jpg)
Whole-Body Disposition and Metabolism of [14C]-2,4,4′-Trichlorobiphenyl (PCB28) Following Lung Administration in Rats
Toxicities of lower-chlorinated biphenyls (LC-PCBs) have drawn increasing attention due to growing evidence of their presence in school indoor air, with 2,4,4′-trichlorobiphenyl (PCB28) being a prevalent congener.

Novel Workflow for Non-Animal PBK Modelling of UV Filters: Oxybenzone as a Case Study
Physiologically based kinetics (PBK) modelling provides (internal) exposure concentrations. We used a PBK model parameterized exclusively with in silico and in vitro data in a bottom-up approach to predict the pharmacokinetics of oxybenzone, a UV filter, present in two formulations (for which dose-normalized Cmax and AUC from clinical studies were different).

Emerging Perspectives on Leveraging Physiologically Based Biopharmaceutics Modeling (PBBM) for BCS Class III Biowaivers: a Webinar Summary
The regulatory framework for Biopharmaceutics Classification System (BCS) class III drug products provides a pathway for streamlined biowaivers in drug development, eliminating the need for expensive...

QSAR-based Physiologically Based Pharmacokinetic (PBPK) Modeling for 34 Fentanyl Analogs: Model Validation, Human Pharmacokinetic Prediction and Abuse Risk Insights
Fentanyl analogs, as emerging new psychoactive substances (NPS), pose a global public health threat due to widespread abuse, high toxicity, and frequent overdose fatalities.

Development of Co-Amorphous Systems for Inhalation Therapy—Part 2: In Silico Guided Co-Amorphous Rifampicin–Moxifloxacin and –Ethambutol Formulations
Tuberculosis (TB) remains a global health challenge due to long treatment durations, poor adherence, and growing drug resistance. Inhalable co-amorphous systems (COAMS) offer a promising strategy for targeted pulmonary delivery...
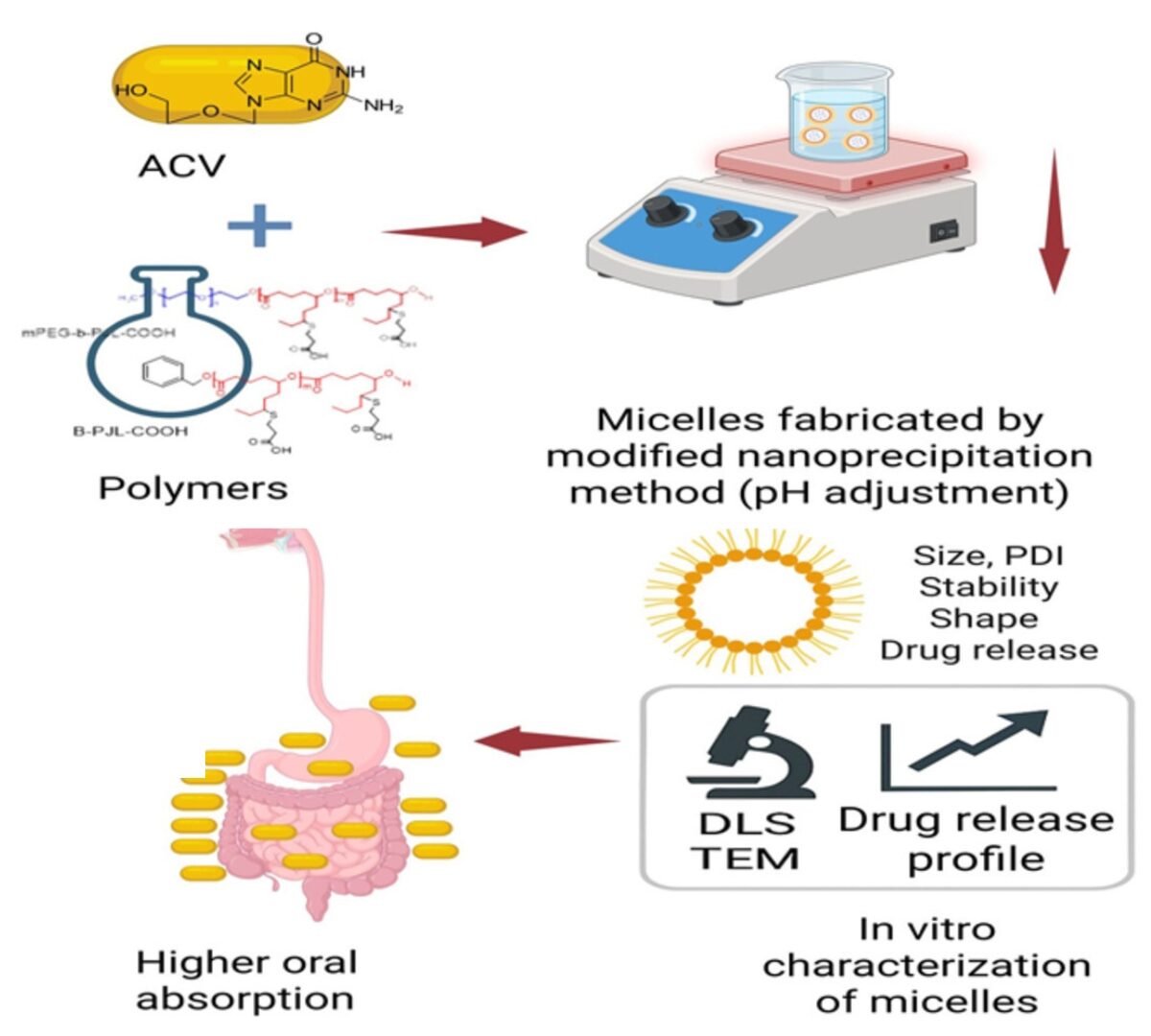
Formulation and Evaluation of Poly (Jasmine Lactone) Based Micelles for Improving the Oral Permeability of Acyclovir
Acyclovir (ACV), an antiviral drug, belongs to the BCS class III drug with intermediate solubility and low permeability.
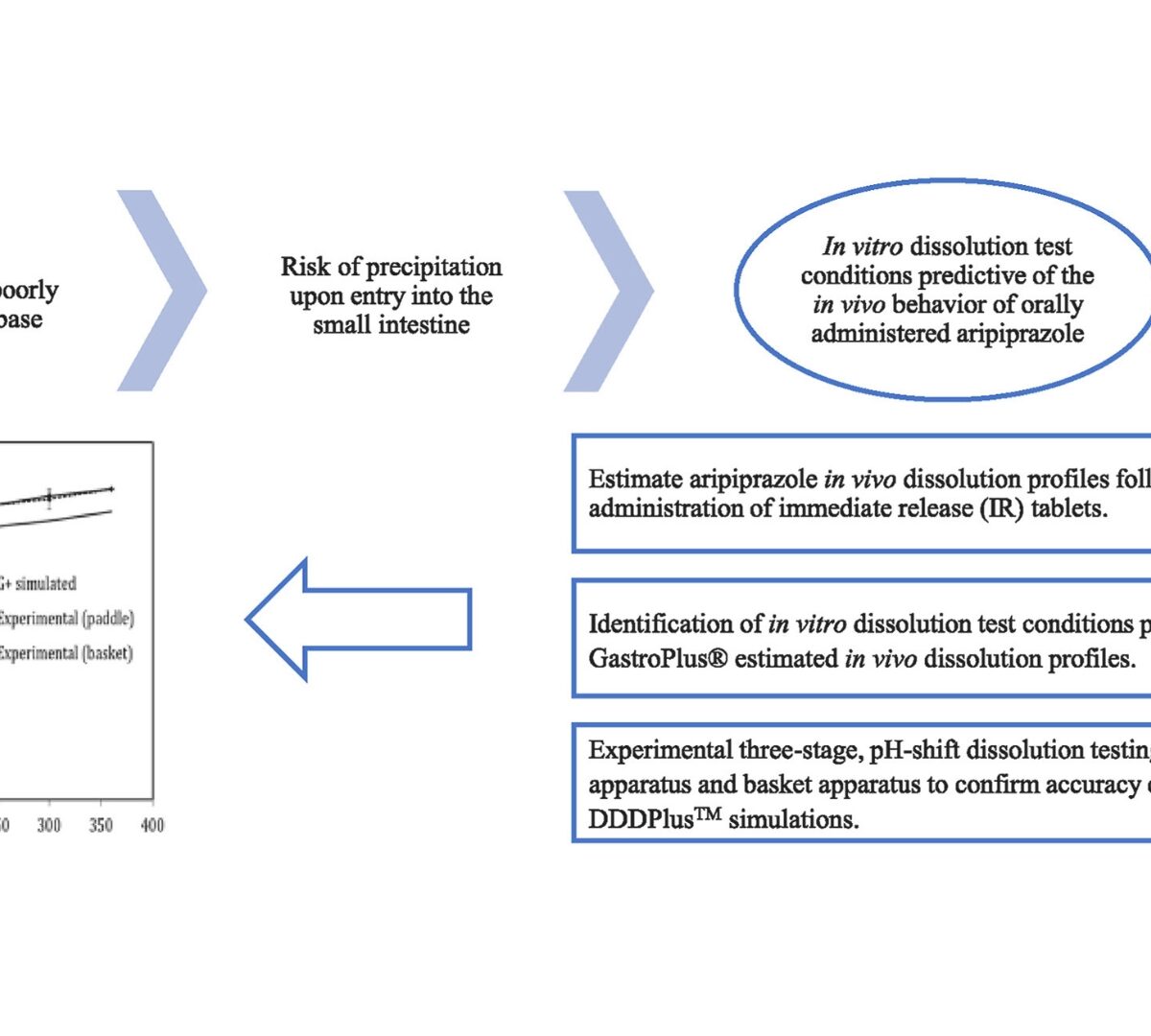
Simulation-Guided Dissolution Testing: Coupling DDDPlus™ and GastroPlus® to Predict Aripiprazole Oral Bioperformance
Orally administered weakly basic compounds like aripiprazole (ARI) can precipitate in the small intestine due to limited solubility at intestinal pH.
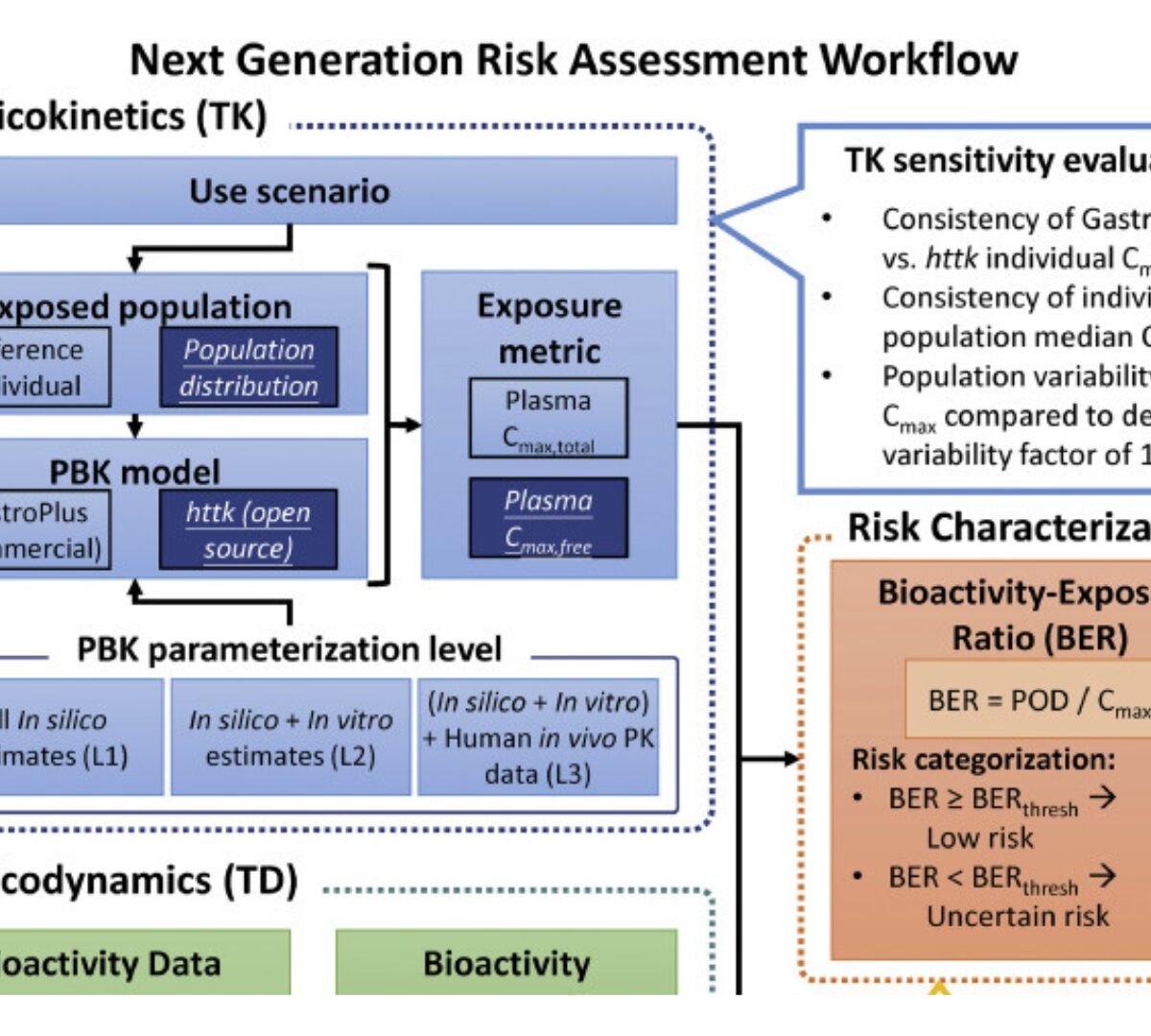
Sensitivity Analysis of the Inputs for Bioactivity-Exposure Ratio Calculations in a NAM-based Systemic Safety Toolbox
To support regulatory decision-making without animal testing, Next-Generation Risk Assessment (NGRA) frameworks leverage New Approach Methodologies (NAM).
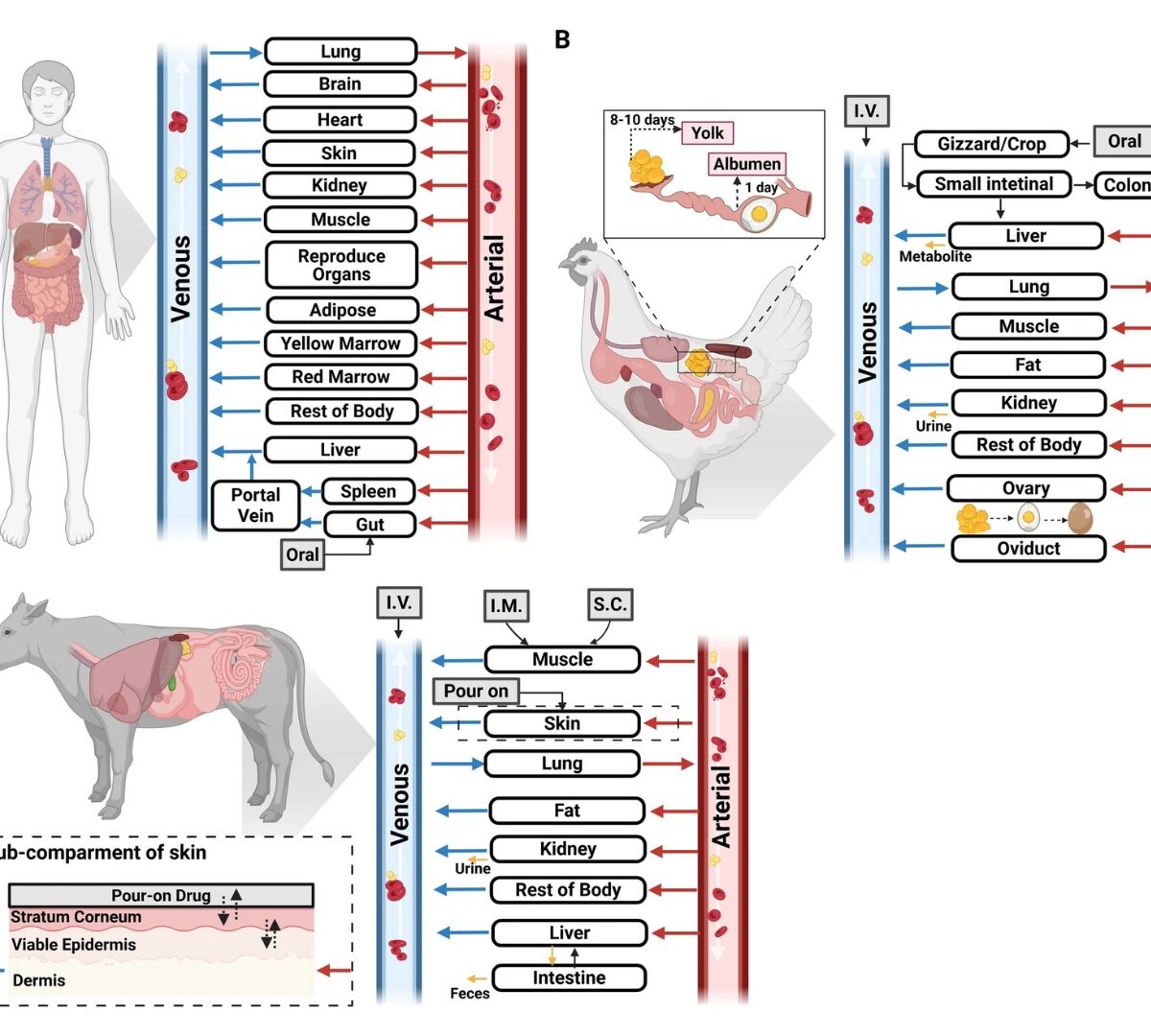
Applications of PBPK Models to Predict Tissue Residues and Extralabel Withdrawal Times of Drugs in Food Animals: Perspectives from the Food Animal Residue Avoidance Databank (FARAD) Program
Physiologically based pharmacokinetic (PBPK) models are commonly used in human drug discovery and development and human health risk assessment of environmental chemicals.
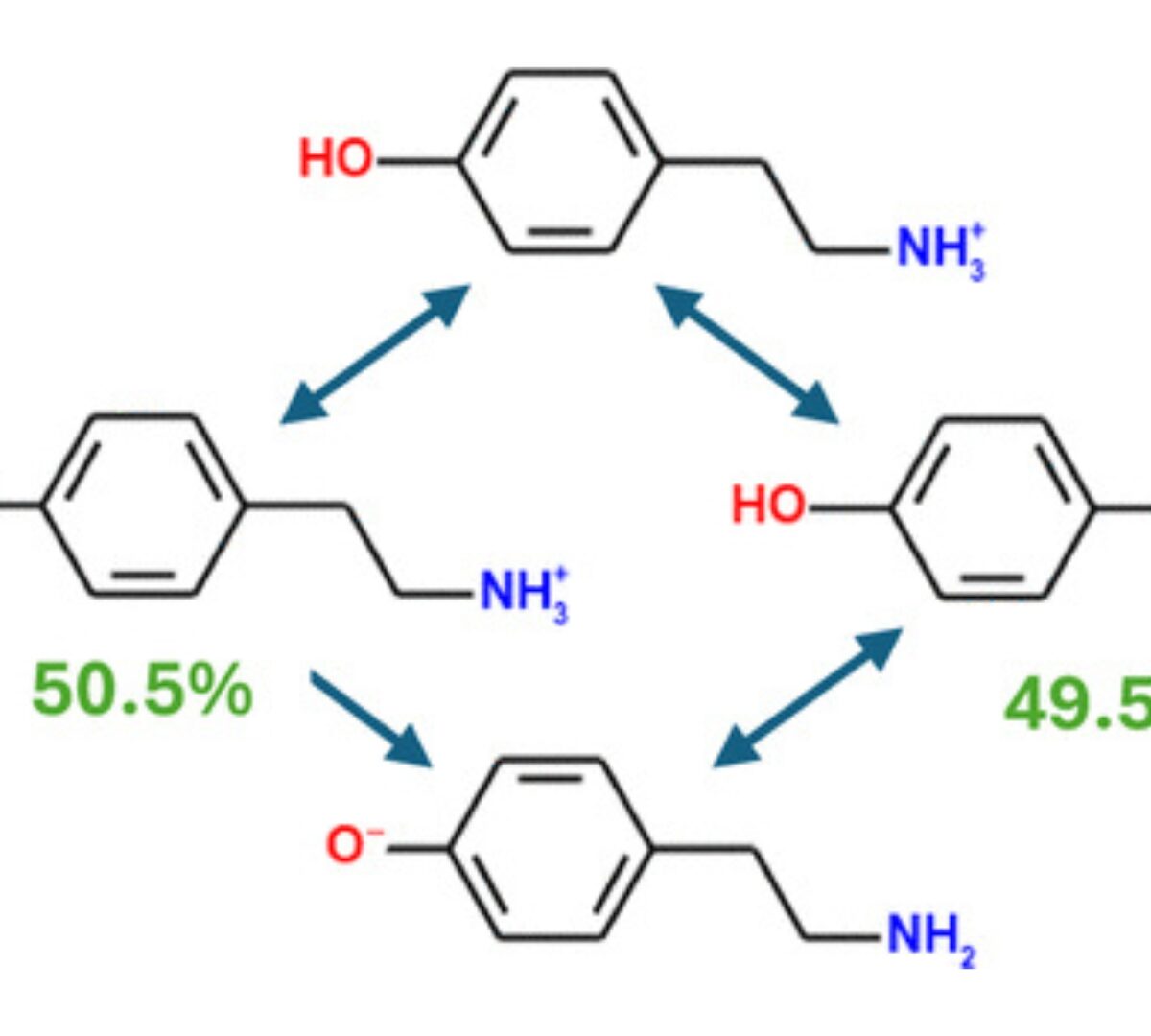
What, This “Base” Is Not a Base? Common Misconceptions about Aqueous Ionization That May Hinder Drug Discovery and Development
The challenges of modern medicinal chemistry increase with the complexity of the chemical compounds studied.
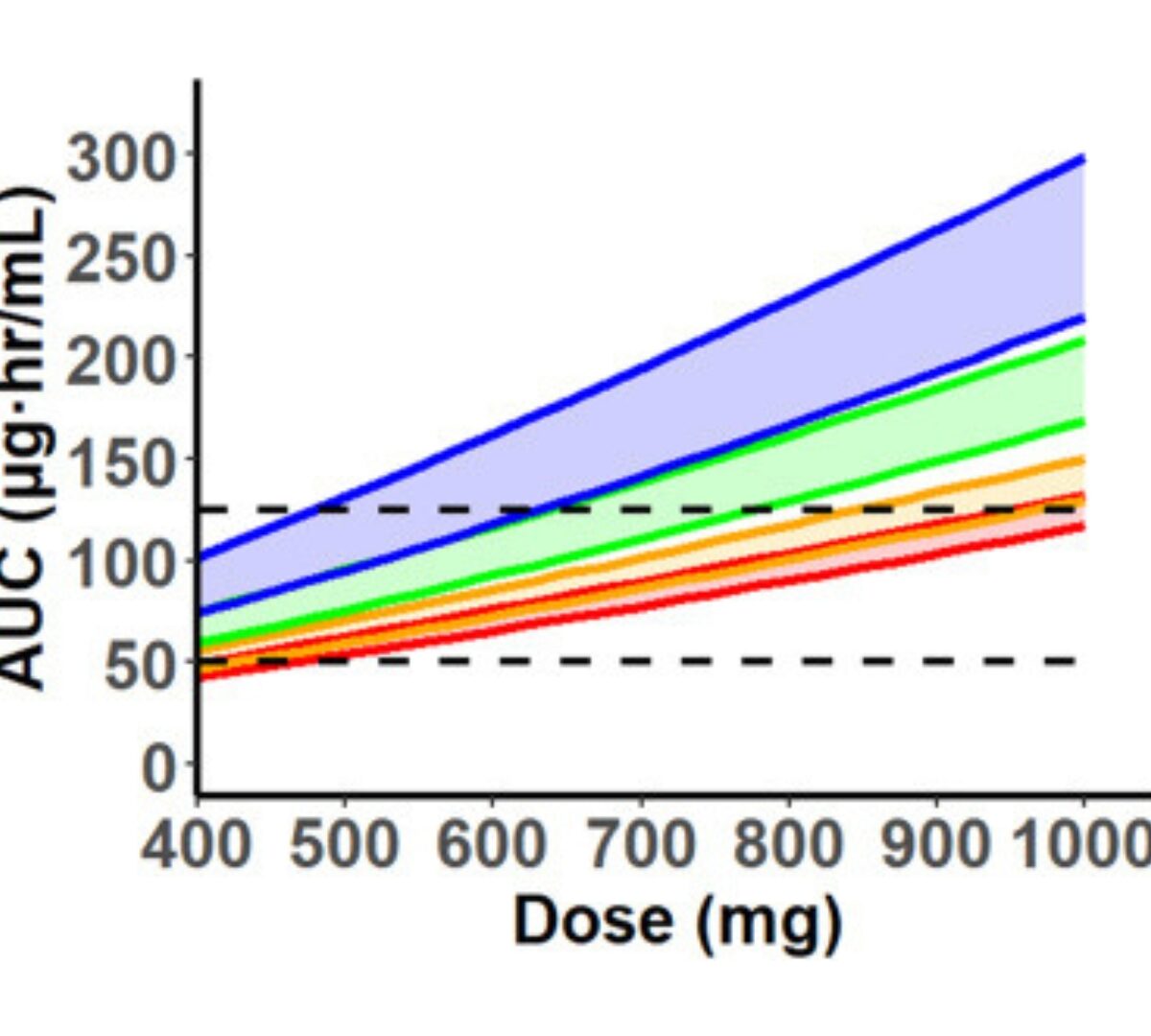
Physiologically Based Pharmacokinetic Modeling of Hydroxyurea: Implications for Dose Adjustment in Patients with Renal Insufficiency
Hydroxyurea is widely used in the management of sickle cell anemia.
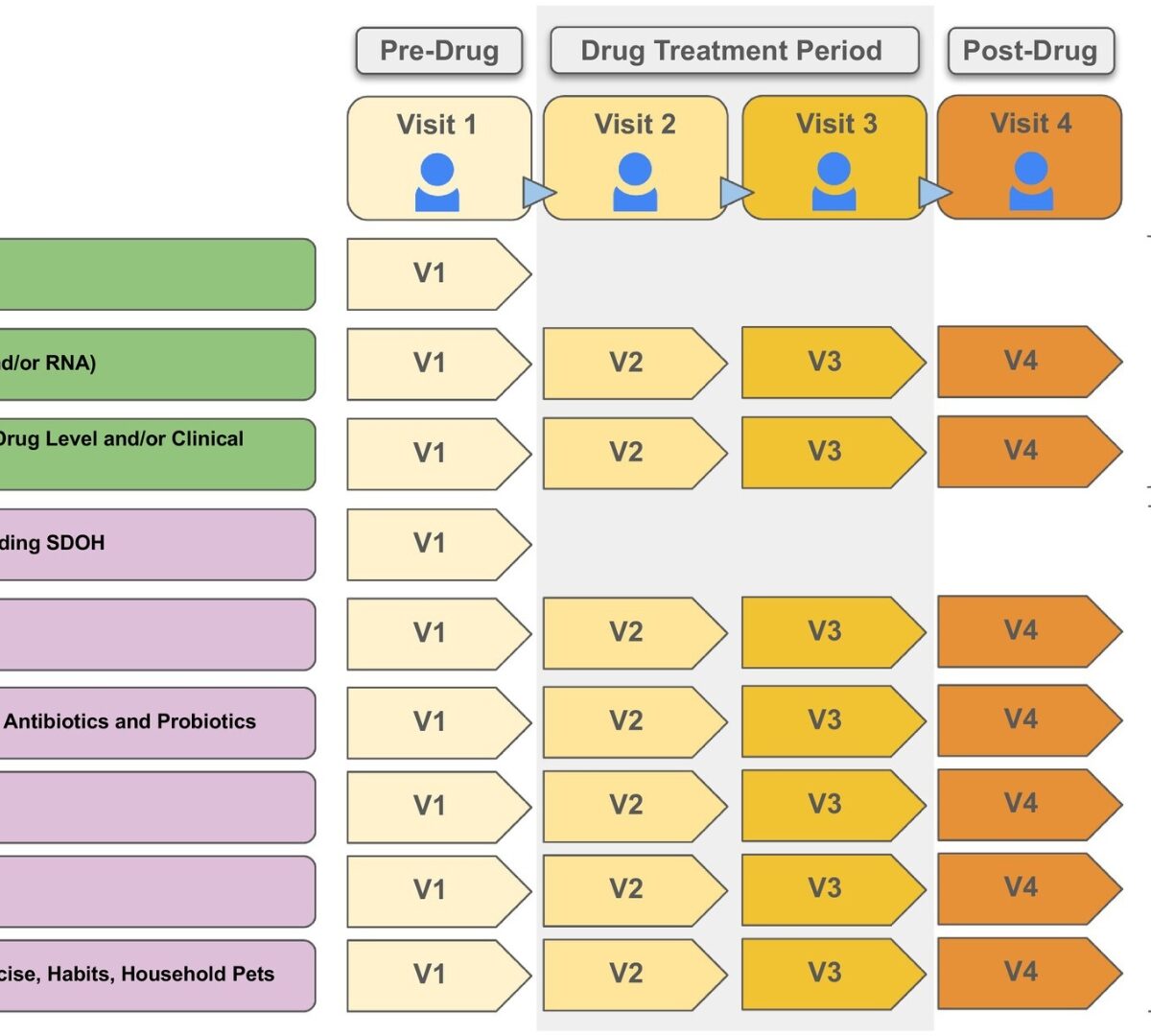
Pharmacomicrobiomics
Oral medications encounter gut commensal microbes that participate directly and indirectly in drug effects through metabolism, interactions with drug metabolites, or production of substrates that compete with drugs for drug-metabolizing enzymes, consequently influencing drug pharmacokinetics.
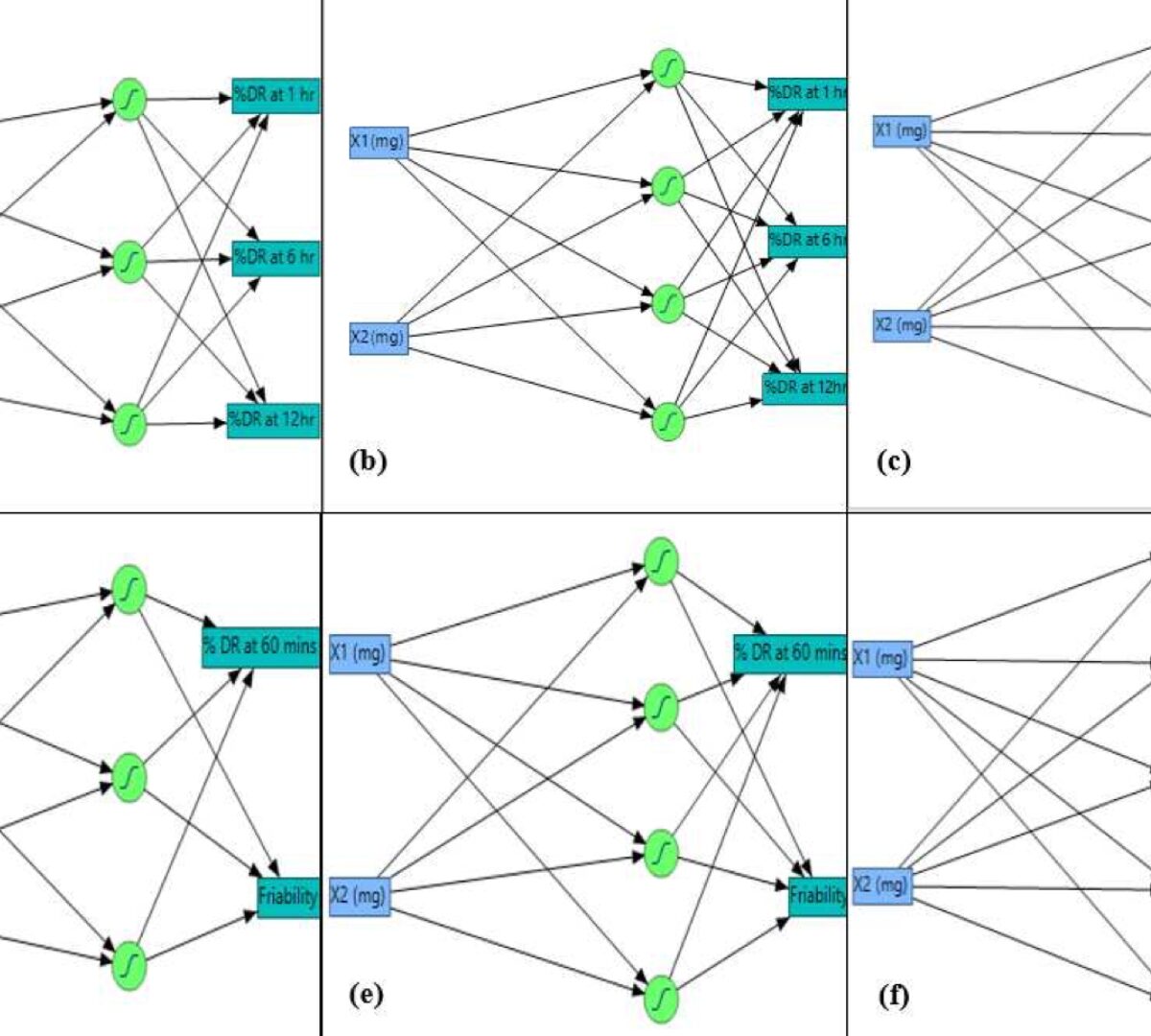
AI-driven ANN and RSM-CCD Integrated Optimization of Cinnarizinedomperidone Bilayer Tablet: In-vitro Evaluation and In-silico PBPK Modeling Using GastroPlus®
Response surface methodology coupled with the design of experiments identifies the optimal response surface function related to selected independent factors.
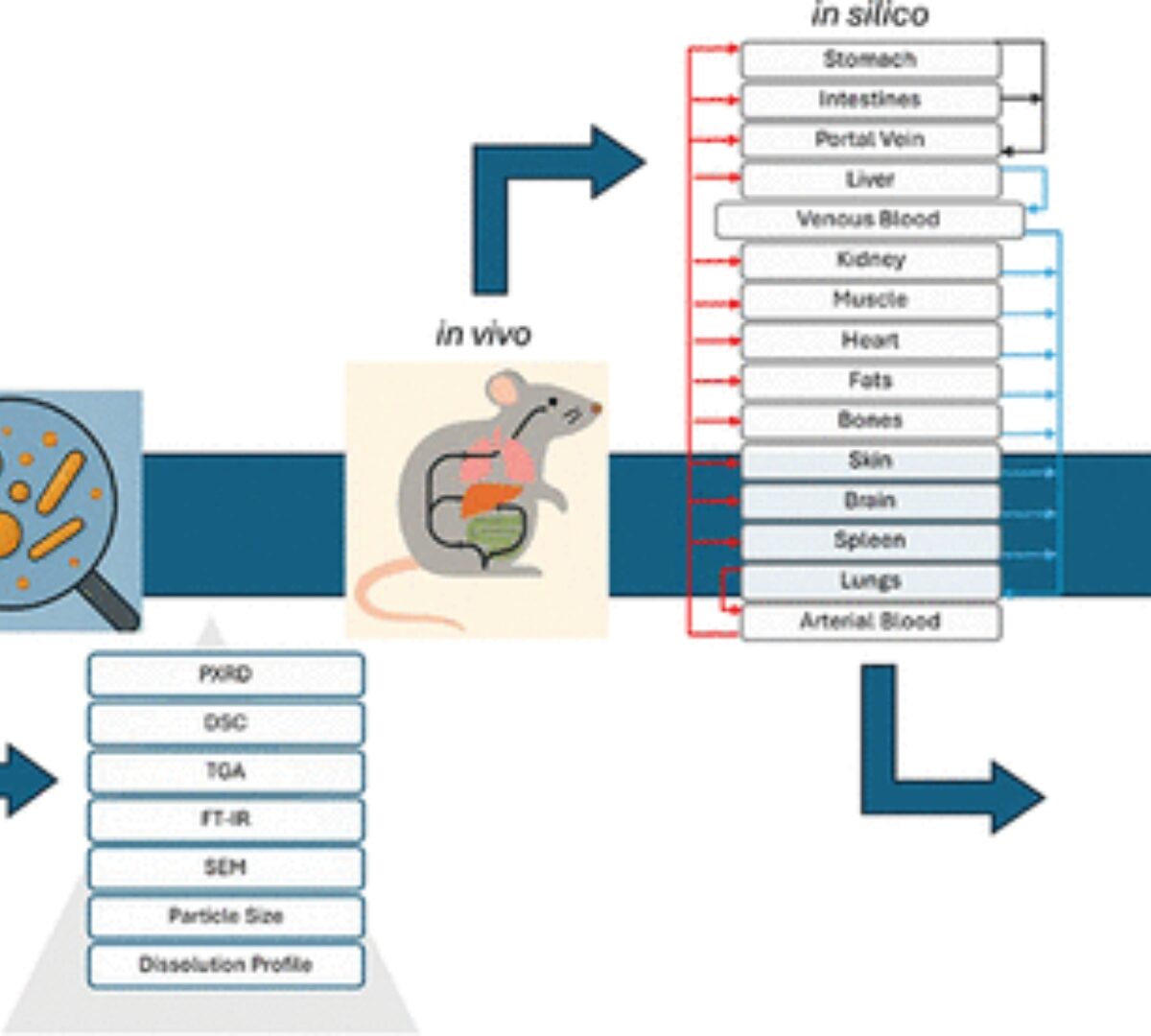
Physiologically Based Pharmacokinetic Modeling of Efavirenz Nanoparticles: from Animal Model to Human Extrapolation
The present work aims to establish a formulation-specific, physiologically based pharmacokinetic (PBPK) model for efavirenz (EFV) nanocrystals that have shown increased dissolution and were produced ...
