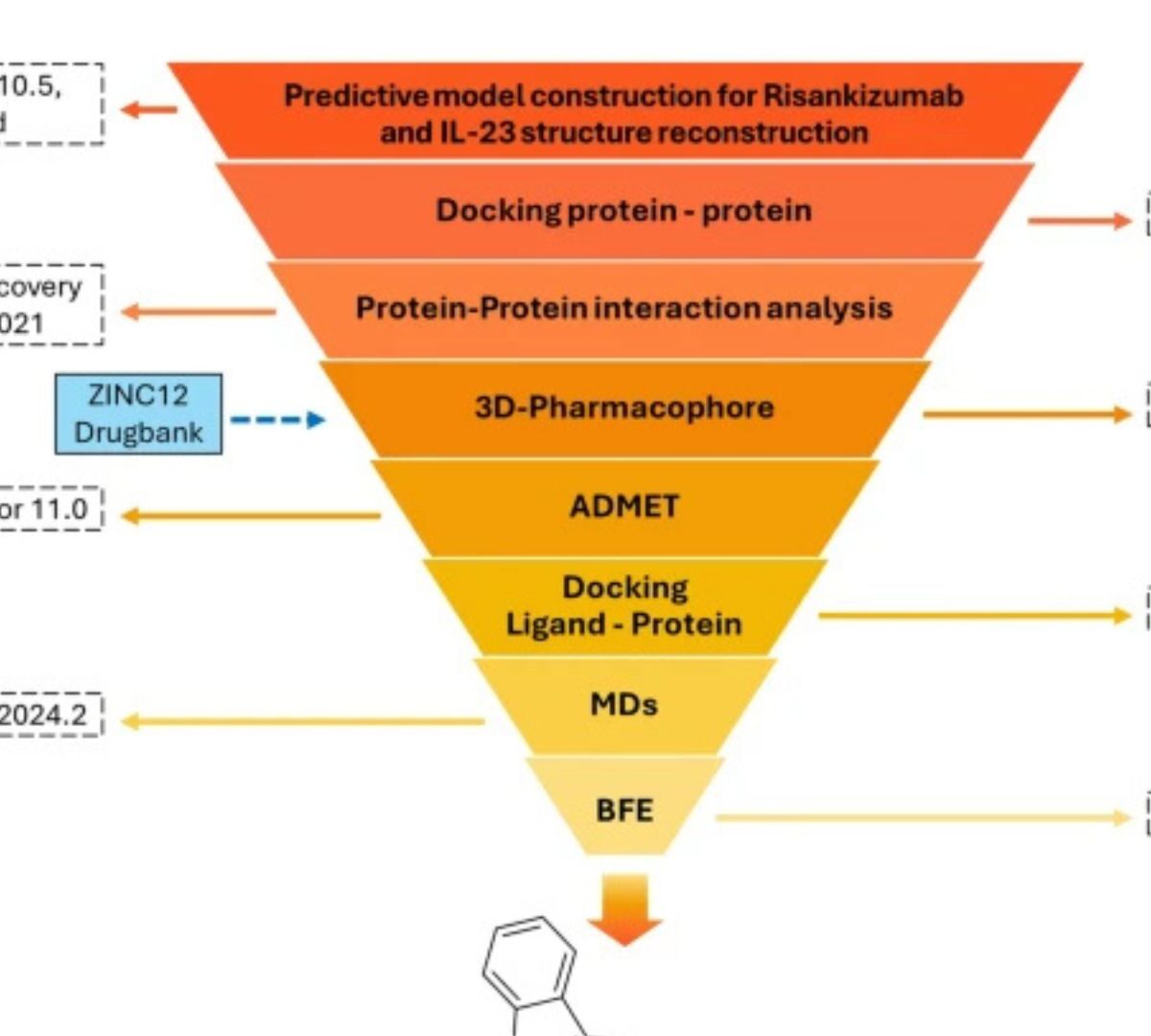
Interleukin-23 (IL-23) is a key driver of chronic inflammatory diseases, yet current therapies rely on costly monoclonal antibodies.
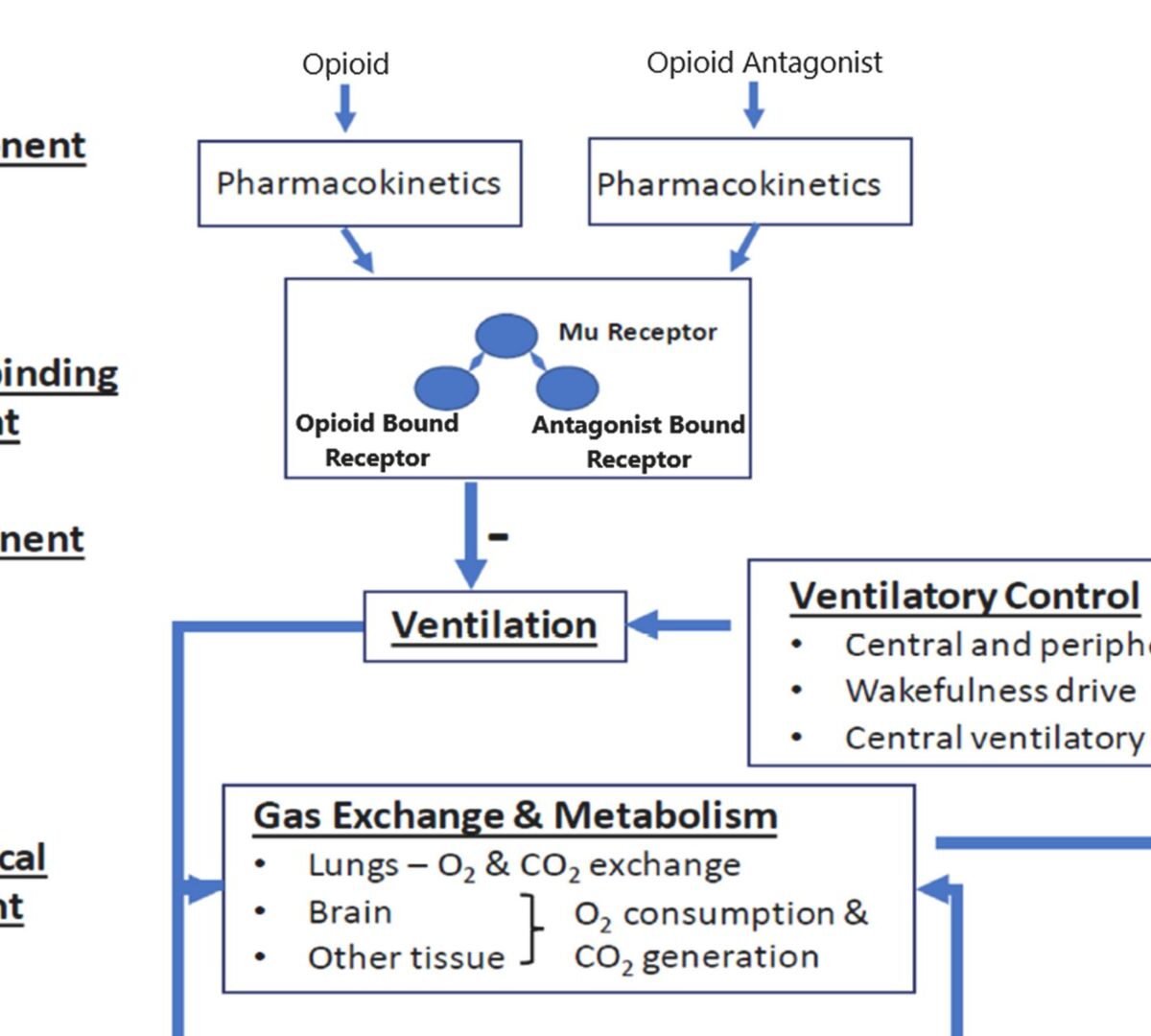
Reversal of a Synthetic Opioid Overdose: Insights From a Validated Translational Model
Synthetic opioids are linked to >90 % of opioid overdose deaths in the United States.
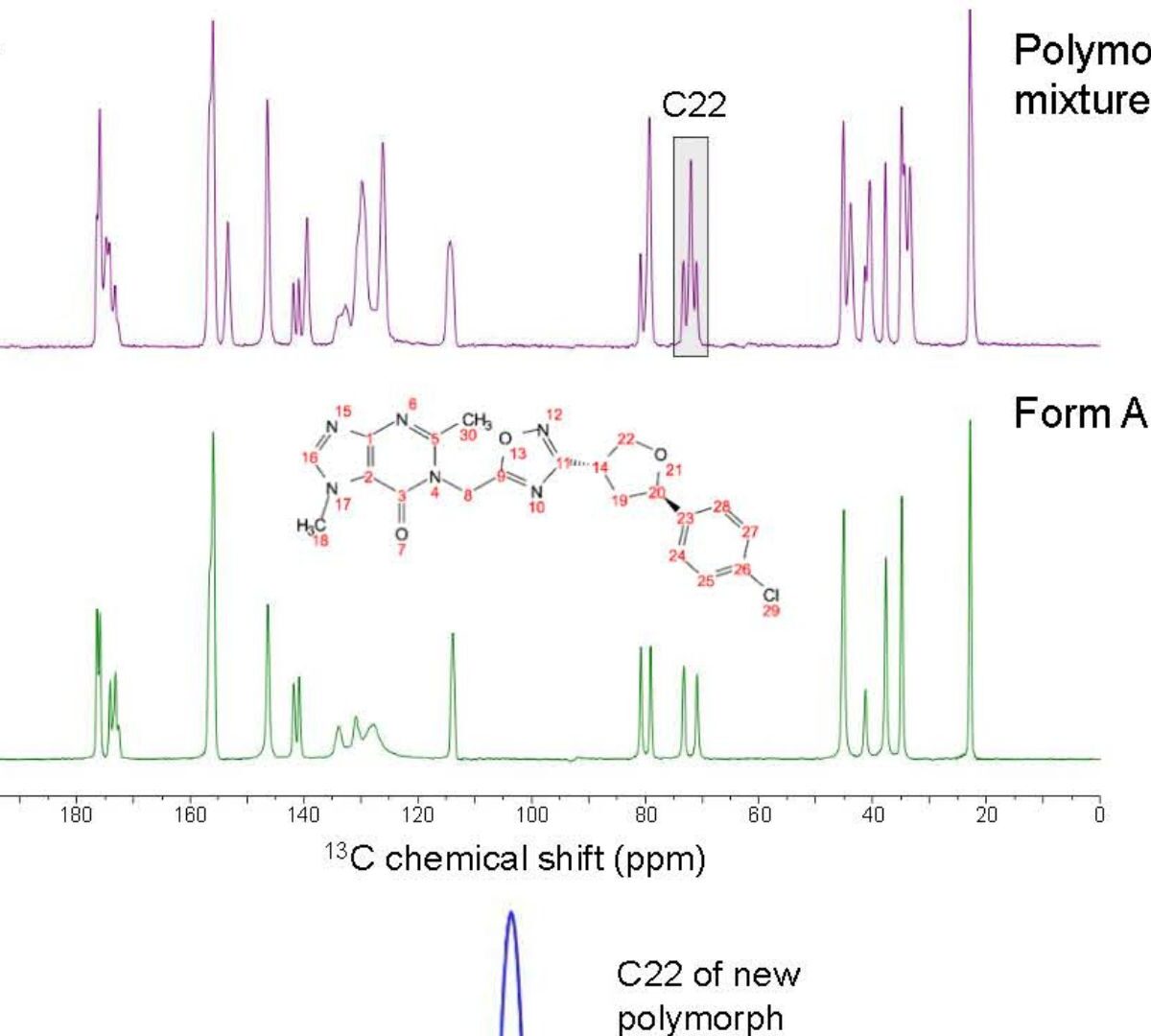
Solid-State Evaluation of a Newly Emerged Polymorph for Early-Stage Pharmaceutical Development
This work presents the solid-state evaluation of a new polymorph (Form M) discovered during the early-stage pharmaceutical development of a new chemical entity GDC-6599.

Identification of a Novel Indolizine RORγT Inverse Agonist Using the AI-Driven Drug Design Platform
Automated multiparameter optimization (MPO) at the point of initial drug design is a powerful emerging approach to improve and expedite drug development.
Establishing Virtual Bioequivalence and Bio-related Dissolution Specifications for Naproxen Using Physiologically Based Pharmacokinetic Modeling and in vitro Biorelevant Dissolution Testing
The aim of the present study was to assess the accuracy of the PBPK model in predicting the pharmacokinetic behavior of weakly acidic BCS class II drugs in humans through a multipronged approach of in vitro dissolution, in vivo studies, and in silico simulations.
Utilizing Metabolism-Based Structure-Activity Relationships and Biokinetic Modeling for Toxicological Evaluation: A Case Study on L-menthyl D-Lactate
Structure activity relationship (SAR) based read across uses existing toxicity data from an analog to predict the toxicity of a target chemical.
![Absorption, Distribution, Metabolism, and Excretion of [14C]SHEN211, a Nonpeptidic Small-Molecule 3CLpro Inhibitor, in Rats](https://www.simulations-plus.com/wp-content/uploads/ChatGPT-Image-Jul-15-2025-at-03_34_17-PM-1200x1024.jpg)
Absorption, Distribution, Metabolism, and Excretion of [14C]SHEN211, a Nonpeptidic Small-Molecule 3CLpro Inhibitor, in Rats
SHEN211 is a selective 3-chymotrypsin-like protease inhibitor that can protect against severe acute respiratory syndrome coronavirus 2.
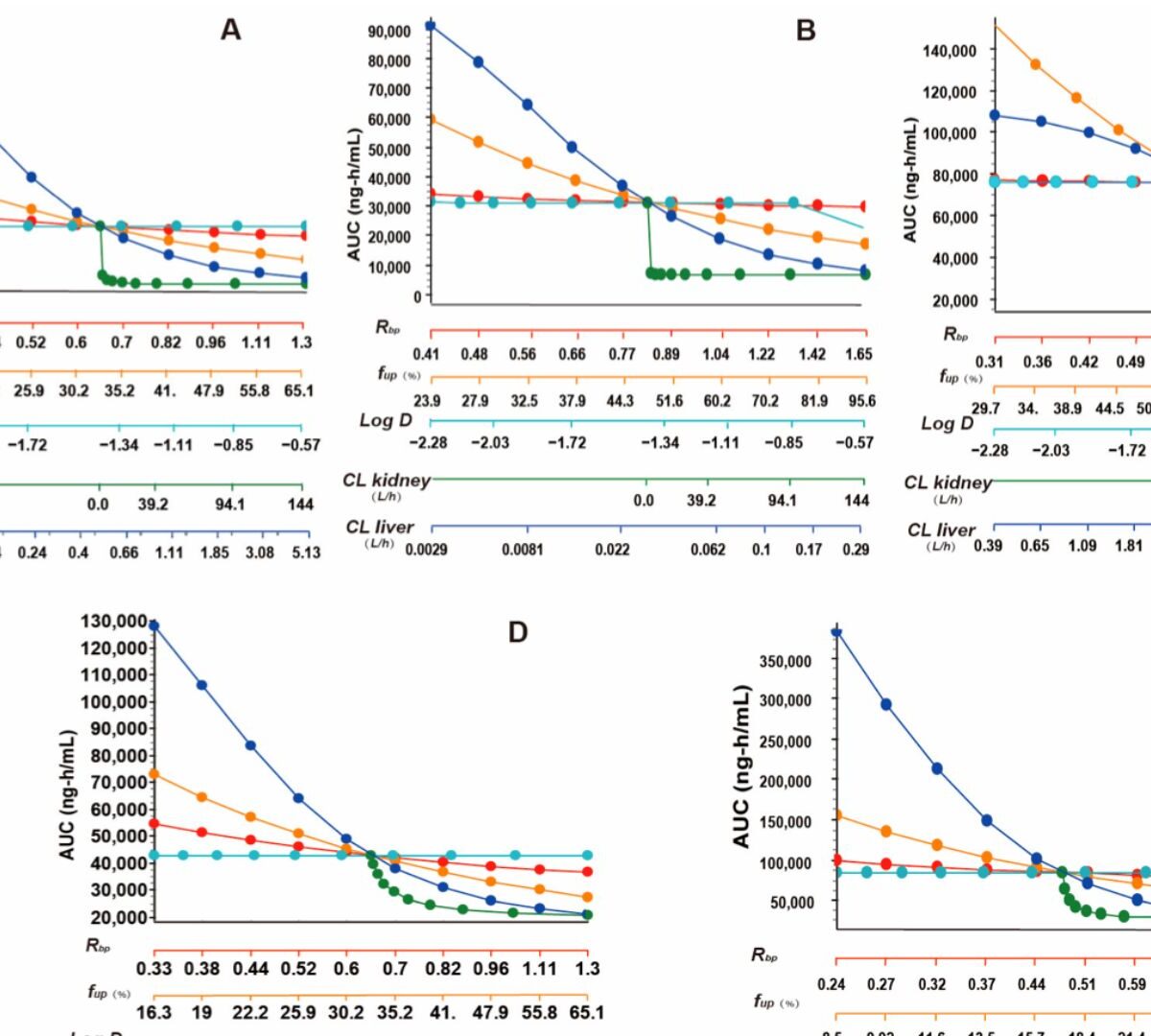
Tissue Distribution and Pharmacokinetic Characteristics of Aztreonam Based on Multi-Species PBPK Model
As a monocyclic β-lactam antibiotic, aztreonam has regained attention recently because combining it with β-lactamase inhibitors helps fight drug-resistant bacteria.
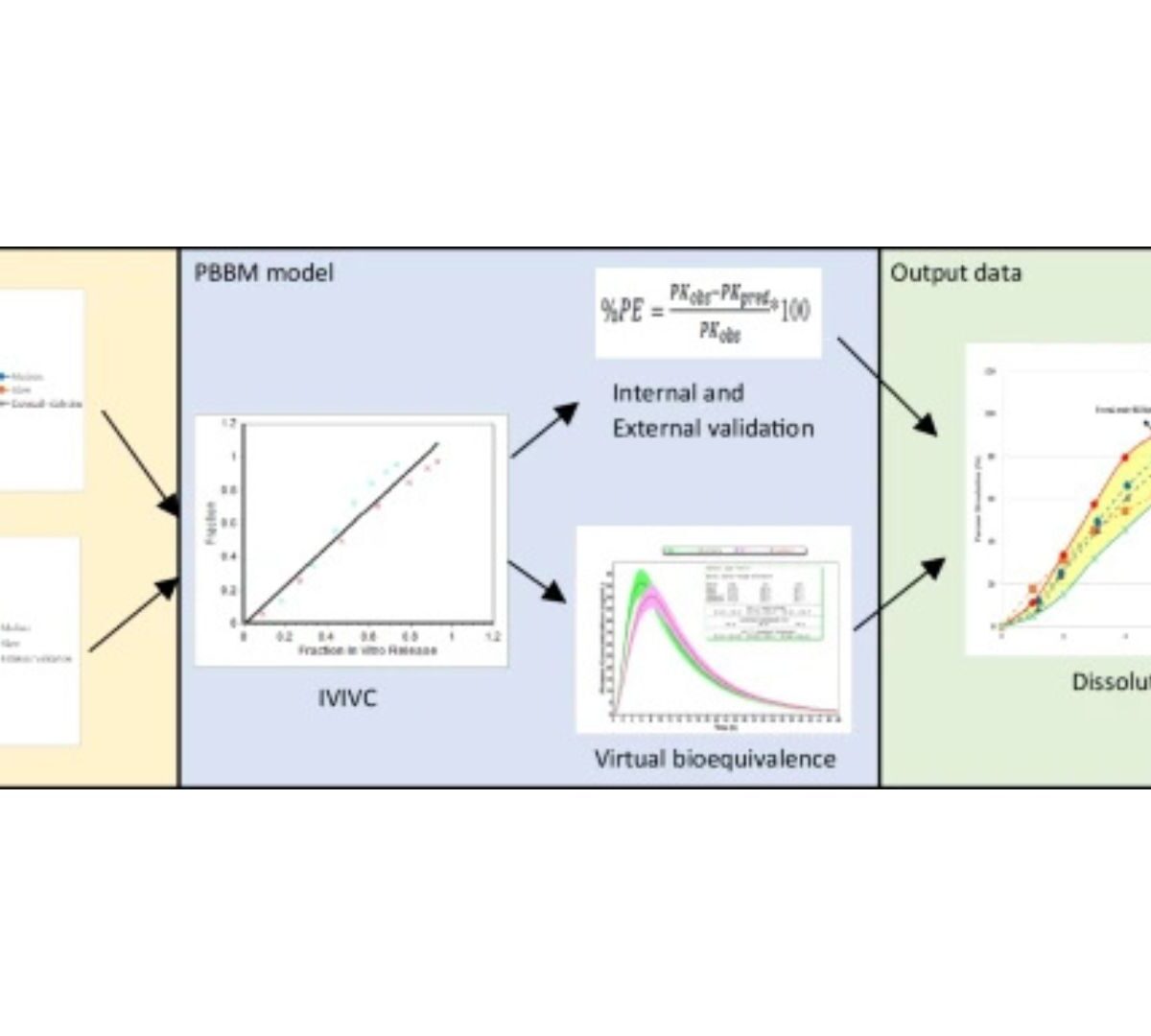
Application of Physiologically Based Biopharmaceutics Modeling (PBBM) to Establish Clinically Relevant Dissolution Specifications for a Prolonged Release Tablet Formulation of Verapamil, a BCS Class I Drug
Our work aimed at setting clinically relevant dissolution specifications for a prolonged release formulation of verapamil, a BCS Class I drug.
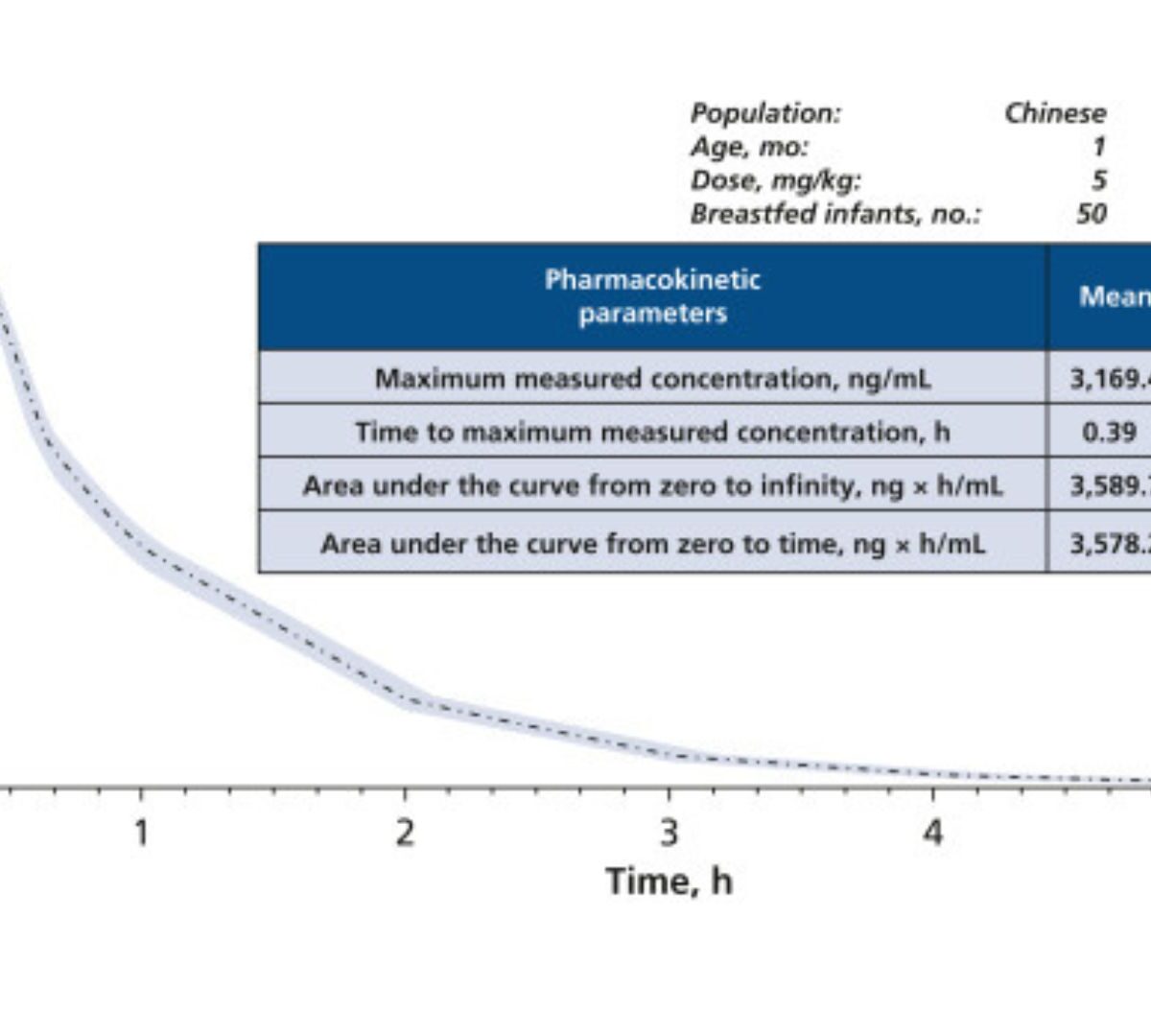
Assessing Whether Breastfeeding is Safe After an Intraoral Injection of 68 mg of Articaine
Limited information is available about the transfer of articaine into breast milk and the associated risks to breastfed infants.

Using Model Master Files to Support Oral Drug Product Development and Regulatory Submissions
This report summarizes the proceedings of Session 2 of the two-day public workshop titled “Considerations and Potential Regulatory Applications for a Model Master File” hosted by the U.S. Food and Drug Administration (FDA) and the Center for Research on Complex Generics (CRCG) on May 2–3, 2024.
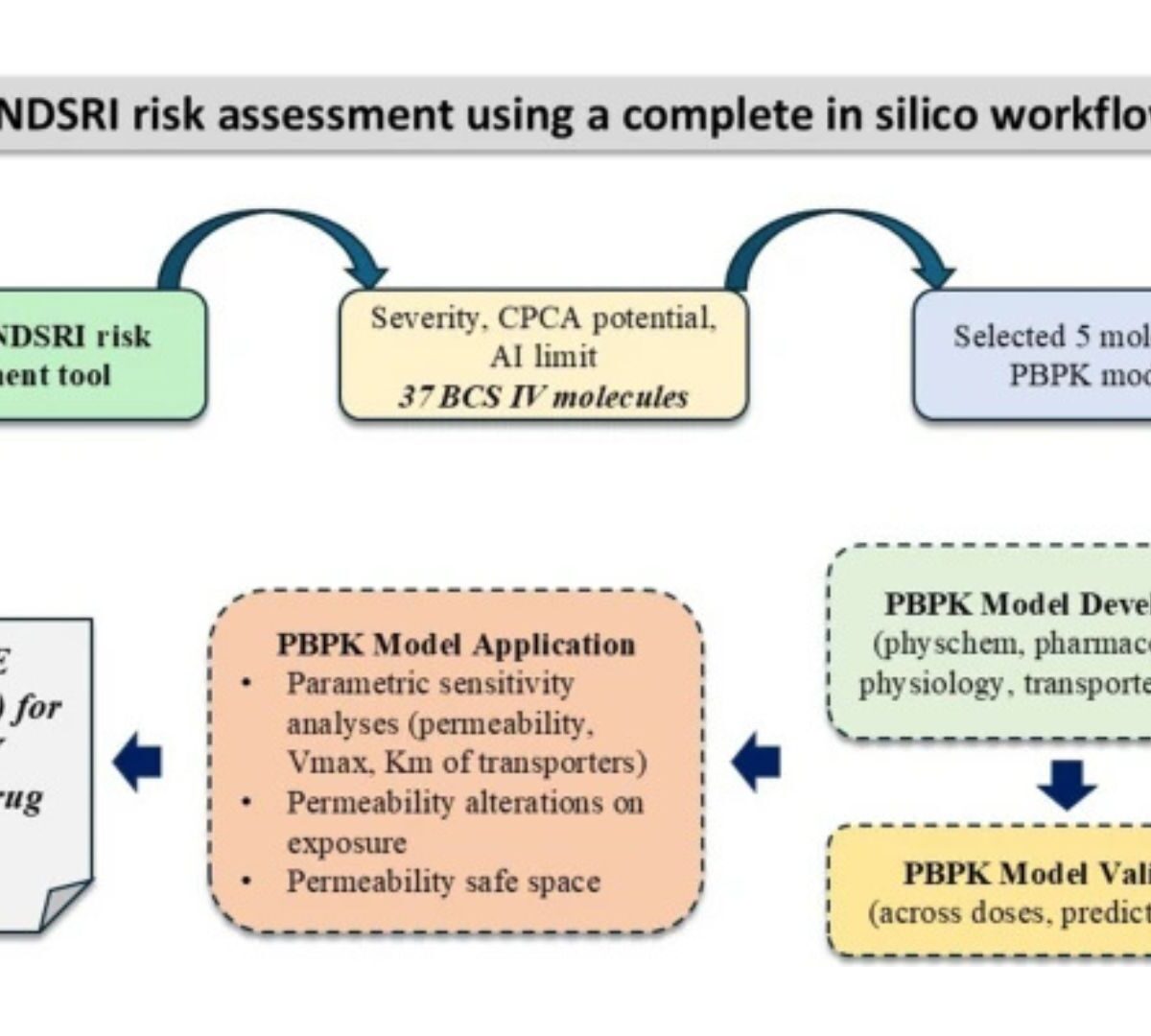
Nitrosamines Risk Assessment for Biopharmaceutics Classification System Class IV Molecule Containing Immediate Release Products: Use of In-Silico Prediction Tools and Physiologically Based Pharmacokinetic Modeling
Nitrosamines drug substance related impurities (NDSRI) are organic impurities, highly potent mutagenic substances that are classified as human carcinogens.
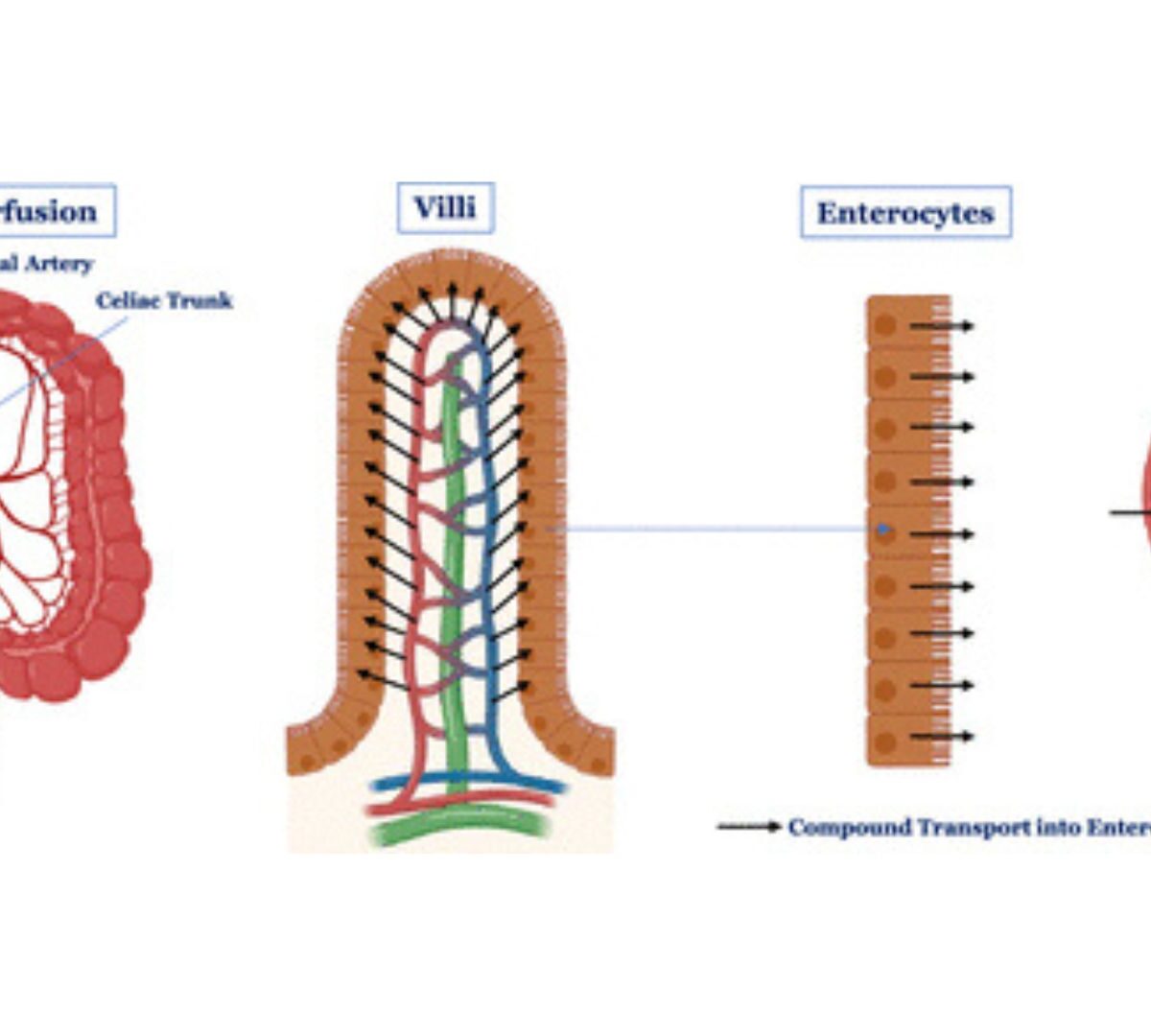
Intestinal Secretion Is a Potentially Important Clearance Mechanism for Low Metabolic Clearance Compounds
Intestinal excretion/secretion (IE) from the systemic circulation via the enterocytes into the intestinal lumen has traditionally been considered a minor clearance (CL) pathway.
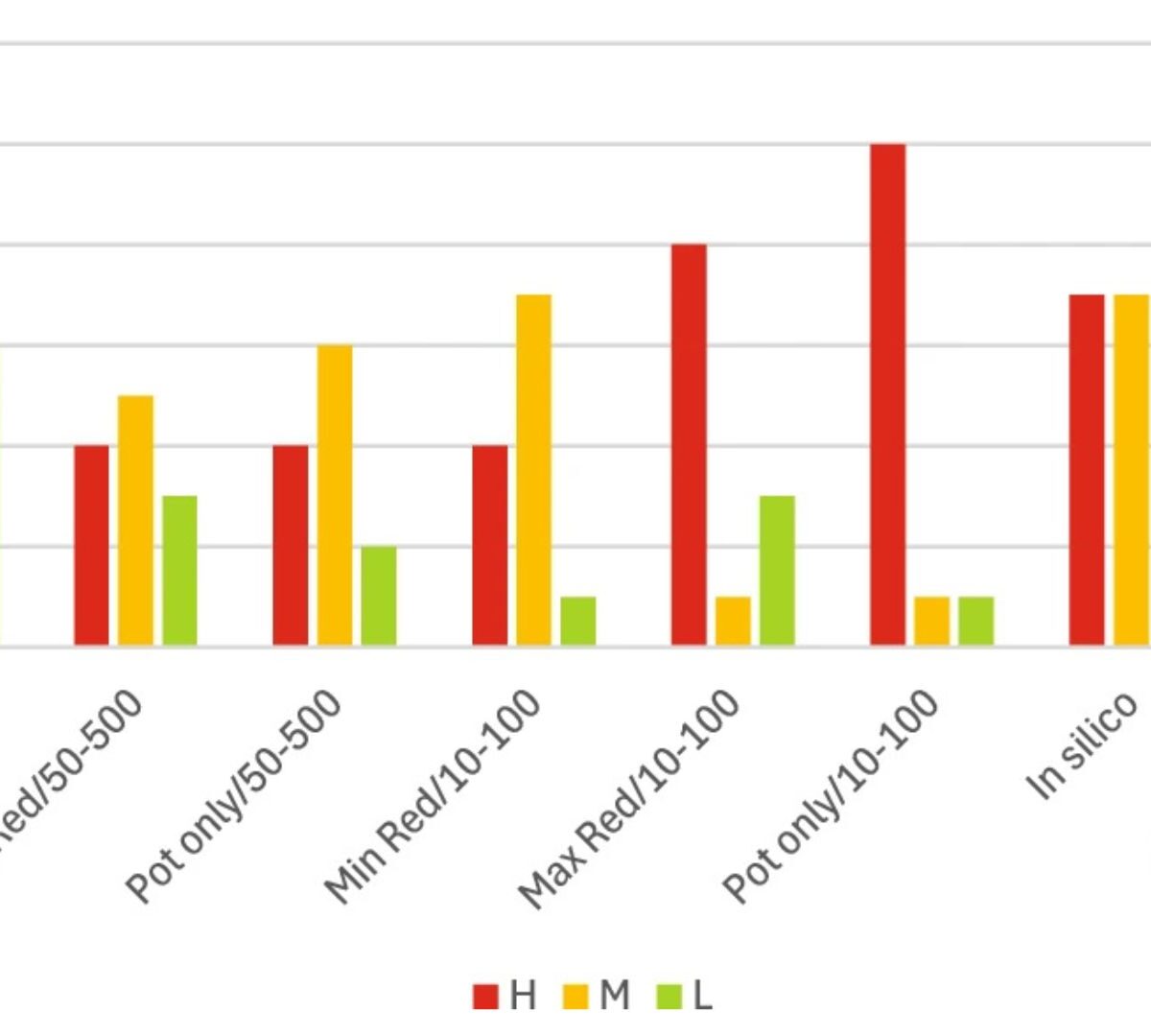
Framework for Classifying Chemicals for Repeat Dose Toxicity Using NAMs
EPAA’s ‘NAM Designathon 2023’ challenge for human toxicity sought to identify a classification system capable of categorising chemicals based on their bioactivity and bioavailability properties determined using non-animal methodologies (Worth et al. 2025).

Multi-target Property Prediction and Optimization Using Latent Spaces of Generative Model
Multi-target property prediction has the potential to improve generalization by exploiting the positive transfer between targets.
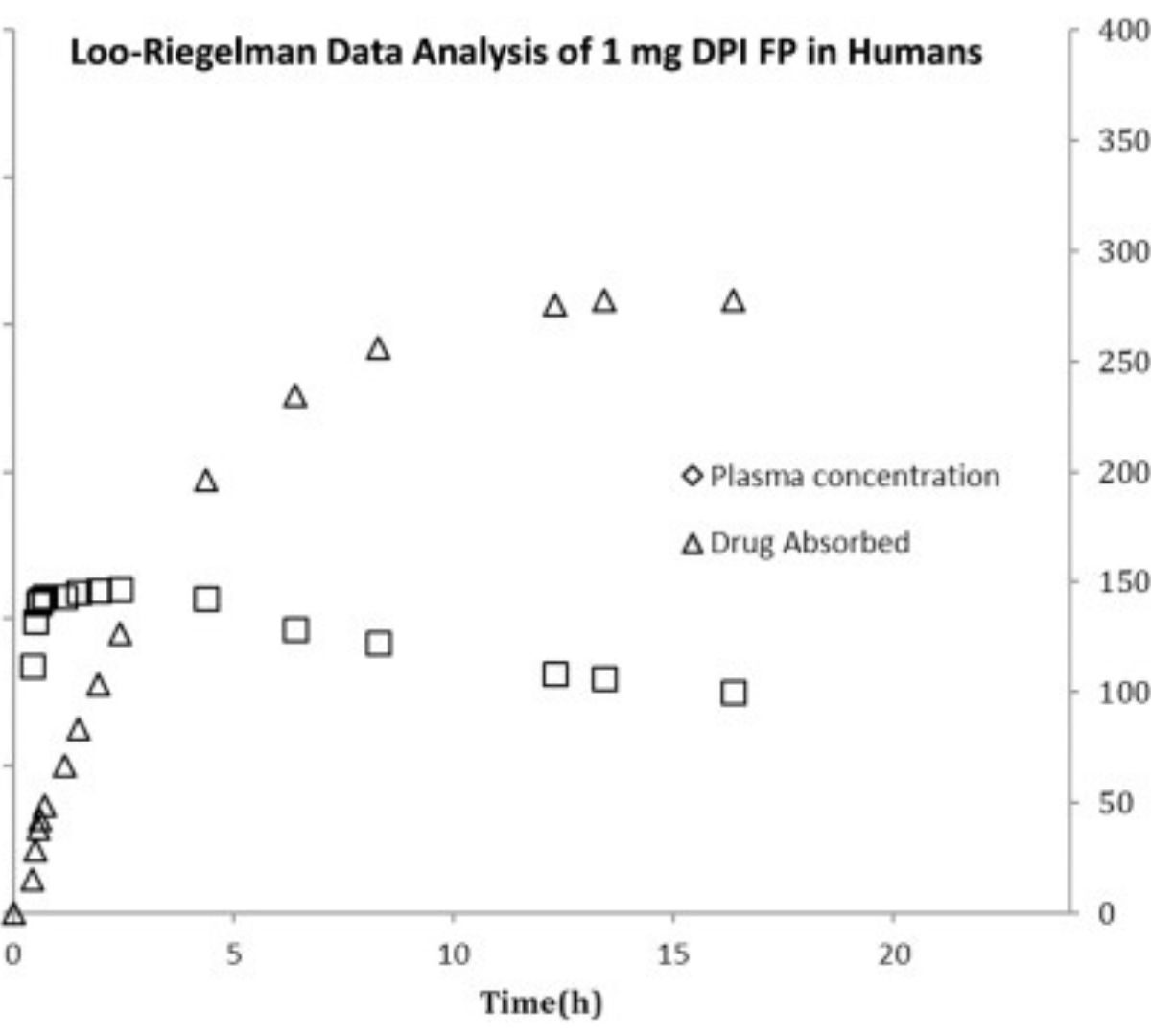
Investigation of the Suitability of Utilizing Plasma Concentration as a Surrogate to Understand Lung Exposure of Inhaled Drug in Rats: Different Delivery Methods of Fluticasone Propionate
Pulmonary diseases, such as asthma and chronic obstructive pulmonary disease (COPD) are complex human airway diseases that affect millions of people worldwide.

From Lab-to-Clinic with Model Informed Formulation Development: a Case Study of Hydroxyzine SR Tablets
Model Informed Formulation Development (MIFD) uses physiologically based pharmacokinetic (PBPK) modelling and other in silico tools to facilitate new product development.
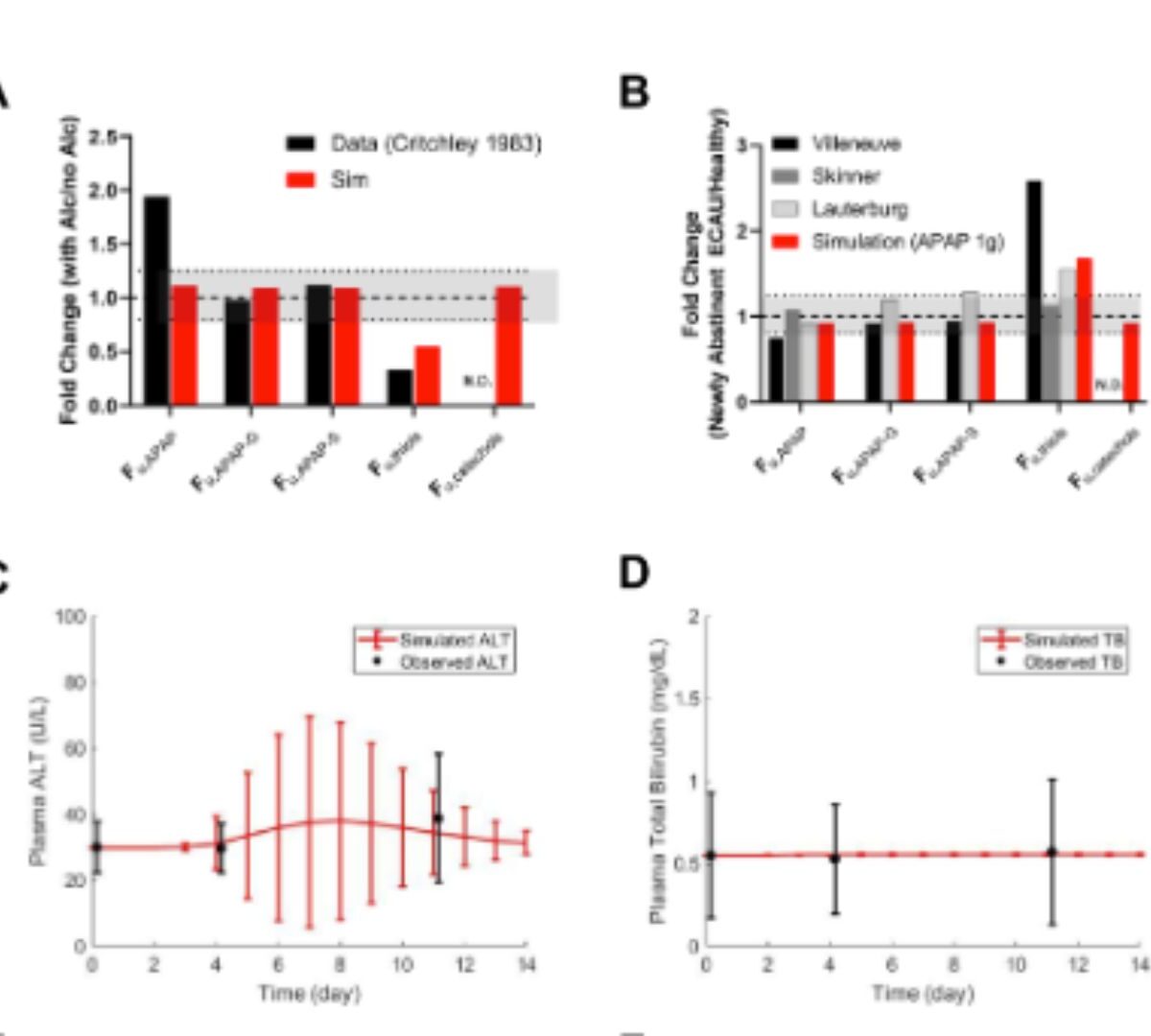
Quantitative Systems Toxicology Modeling of Acetaminophen Pharmacokinetics and Hepatic Biomarkers After Overdoses of Extended-Release and Immediate-Release Formulations in Adults With Chronic Alcohol Use or Low Glutathione
Acetaminophen (APAP), an over-the-counter analgesic and antipyretic, can cause hepatotoxicity when ingested in large overdoses.
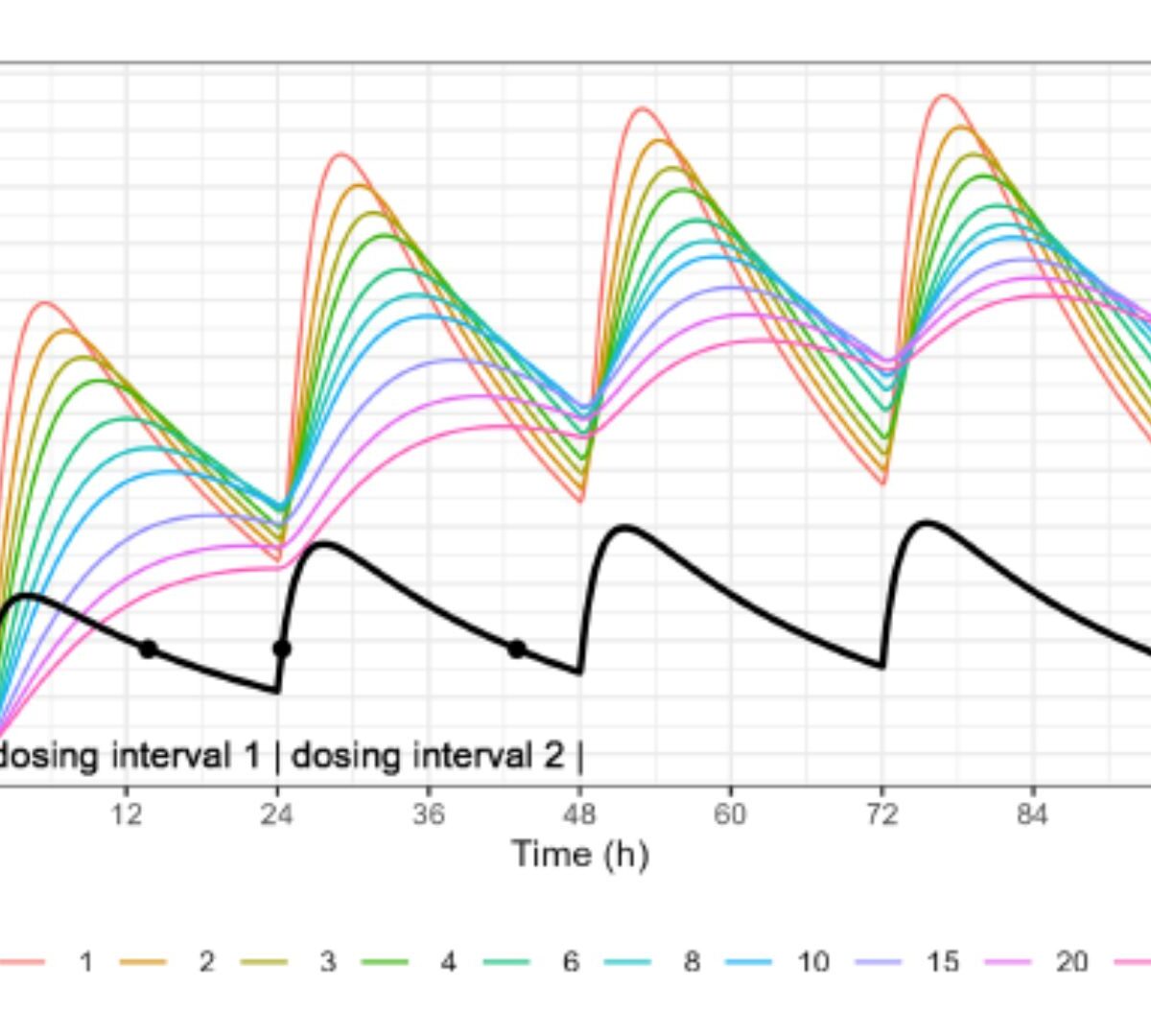
Beyond the Linear Model in Concentration-QT Analysis
This work introduces several extensions for concentration-QT modeling in a pharmacometric context.
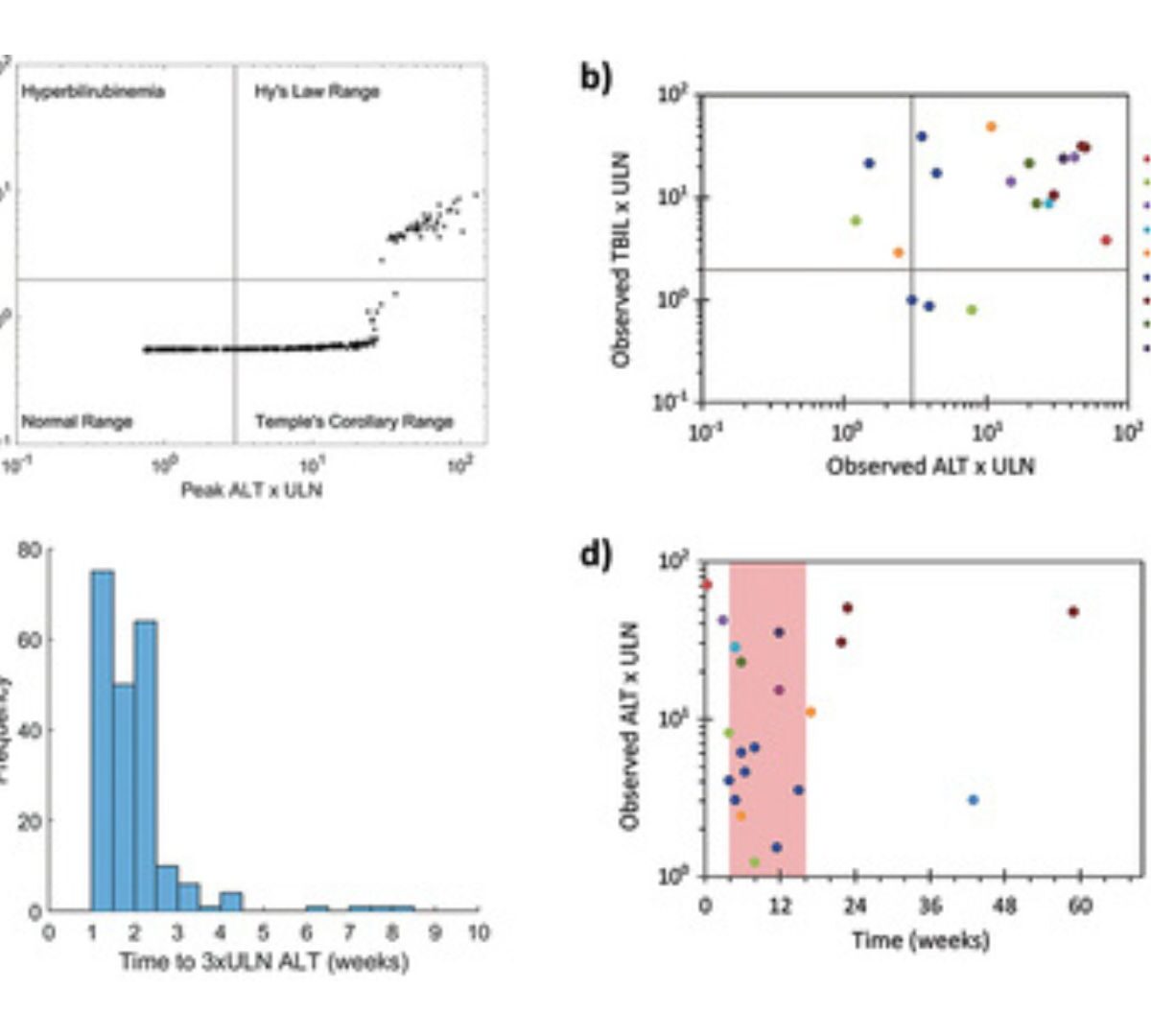
A Well-Characterized Mechanistic Model for Exploring Known or Hypothesized T cell Mediated Drug Induced Liver Injury: Current Capabilities and Challenges for Future Predictivity
Drug-induced liver injury (DILI) is an adverse event whose emergence can slow or halt drug development programs.