Kacie L. Beatch, Ph.D.
Senior Program Manager
Simulations Plus, Inc.
About Kacie
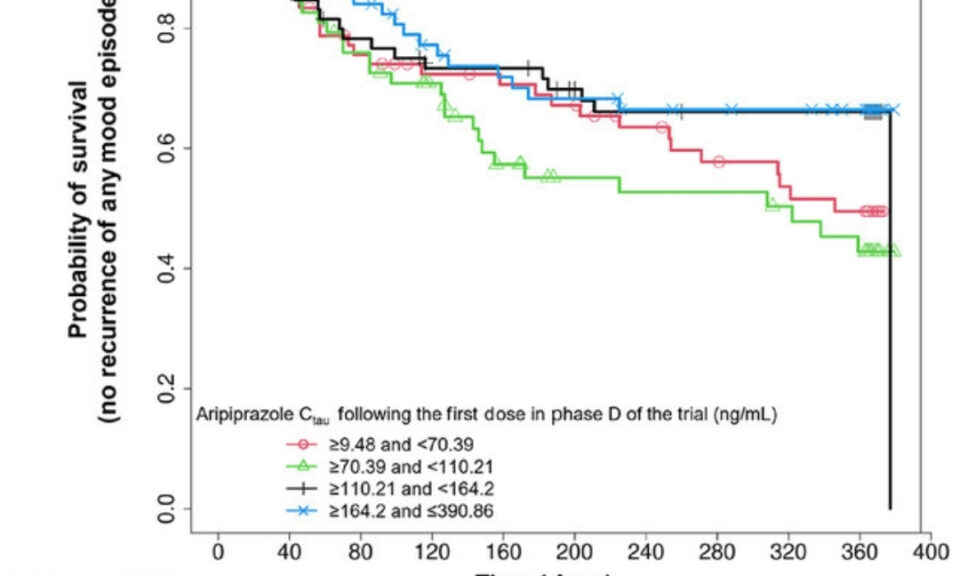
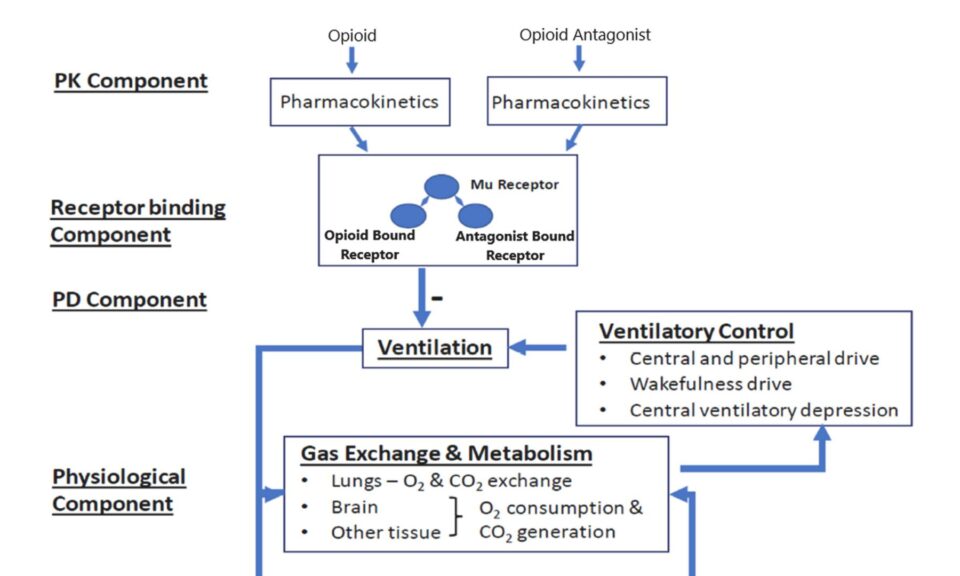
Kacie joined Simulations Plus in May 2020 as Senior Program Manager. She holds a Bachelor of Science degree in Clinical Laboratory Science and a PhD in Pharmaceutical Sciences from North Dakota State University. After a two-year post-doctoral fellowship in Clinical Research, she spent seven years as a Clinical Research Scientist with focus on Phase 1 and generic drug development—contributing to over 40 product development programs and at least two dozen approved FDA submissions. Kacie is a member of the American Association of Pharmaceutical Scientists, the American Society of Clinical Pharmacology and Therapeutics, and maintains her certification as a Medical Laboratory Scientist under the American Society of Clinical Pathology.