Expanding our Immunology and Oncology Drug Development Capabilities

Integrated in vitro – in vivo – in silico studies in the pharmaceutical development of propranolol hydrochloride mucoadhesive buccal films
In order to exploit the advantage of drug delivery through the buccal mucosa, mucoadhesive buccal films with propranolol hydrochloride based on polyethylene oxide...

Consult & Coach Comparison Flyer
Accelerate your project goals with a free 3-month license to Monolix and/or GastroPlus
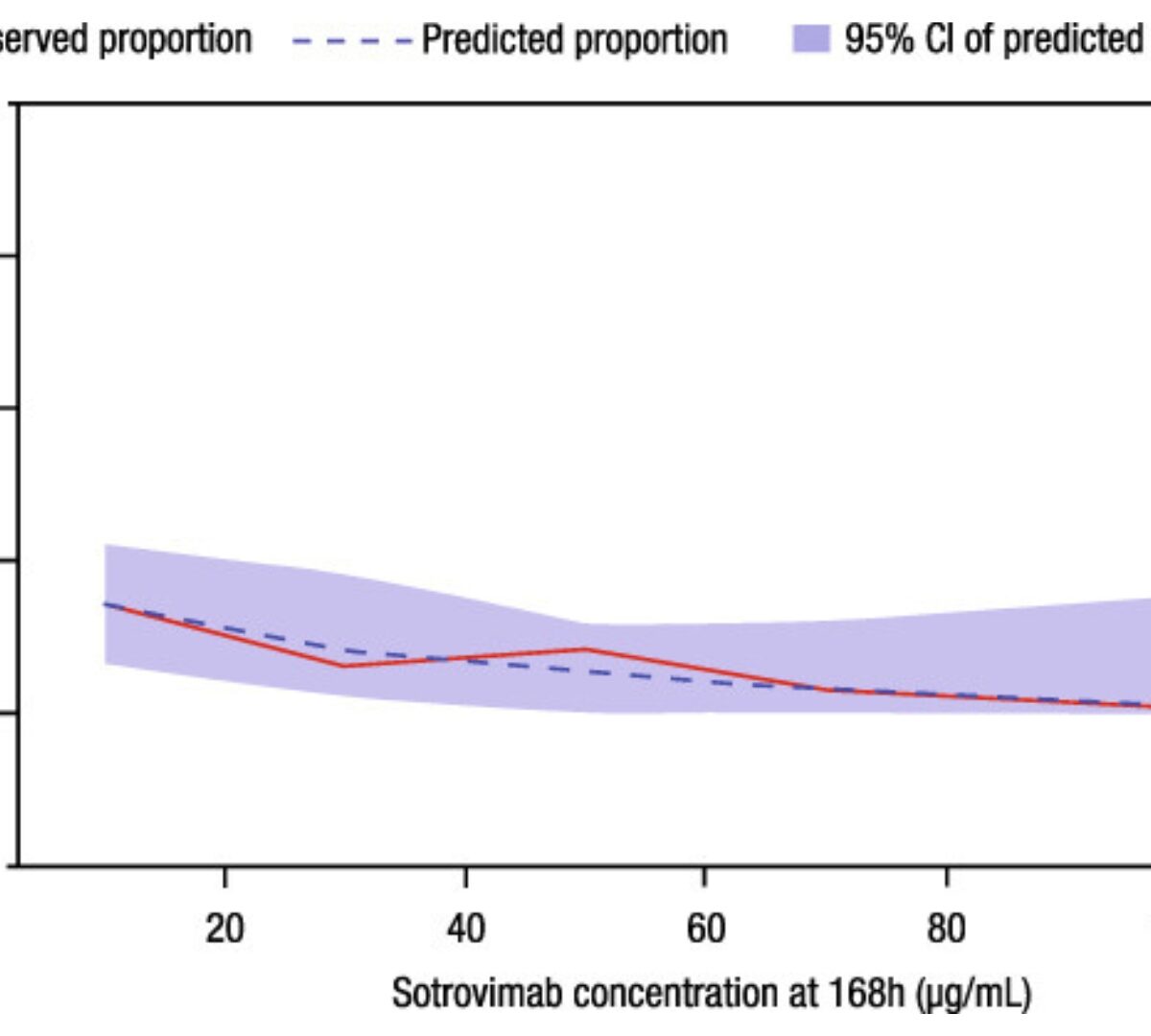
Population Pharmacokinetics and Exposure-Response Analysis of a Single Dose of Sotrovimab in the Early Treatment of Patients With Mild to Moderate COVID-19
Sotrovimab is a recombinant human monoclonal antibody that has been shown to prevent progression to hospitalization or death in non-hospitalized high-risk patients with mild to moderate...

Population pharmacokinetics of vadadustat, a hypoxia-inducible factor prolyl hydroxylase inhibitor for treatment of anemia associated with chronic kidney disease
Vadadustat is an oral hypoxia-inducible factor prolyl hydroxylase inhibitor, a class of...
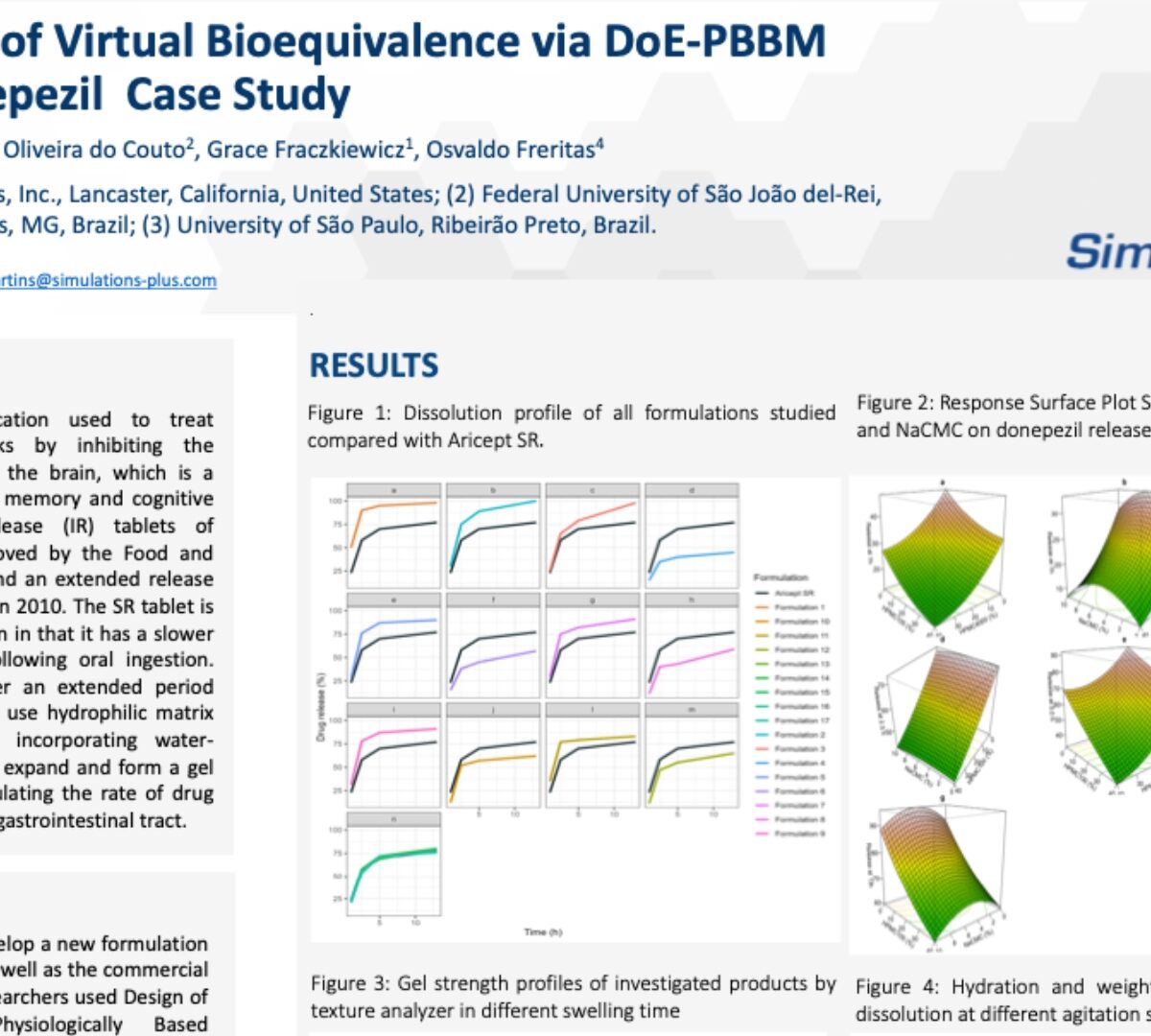
Establishment of Virtual Bioequivalence via DoE-PBBM Model: A Donepezil Case Study
Donepezil (DZP) is a medication used to treat Alzheimer's disease.

ADMET Predictor® 11 is Here: AI & ML for Drug Discovery and Development
Your competitors are using AI and machine learning for ADMET prediction—are you?

AI-Driven Drug Design (AIDD) Flyer
Drug Design, Meet PBPK

Early Drug Discovery (EDD) Flyer
Unleash the Power of AI for Early Drug Discovery

Revisiting the in-vitro and in-vivo considerations for in-silico modelling of complex injectable drug products
Complex injectable drug products (CIDPs) have often been developed to modulate the pharmacokinetics along with efficacy for therapeutic agents used for remediation of chronic disorders.

Modeling Based Approaches to Support Generic Drug Regulatory Submissions-Practical Considerations and Case Studies
Model informed drug development (MiDD) is useful to predict in vivo exposure of drugs during various stages of the drug development process. This approach employs a variety of quantitative tools to assess the risks during the drug development process.

Simulations Plus Acquires Immunetrics to Expand its Immunology and Oncology Drug Development Capabilities
Acquisition increases breadth and depth of QSP expertise and range of therapeutic applications
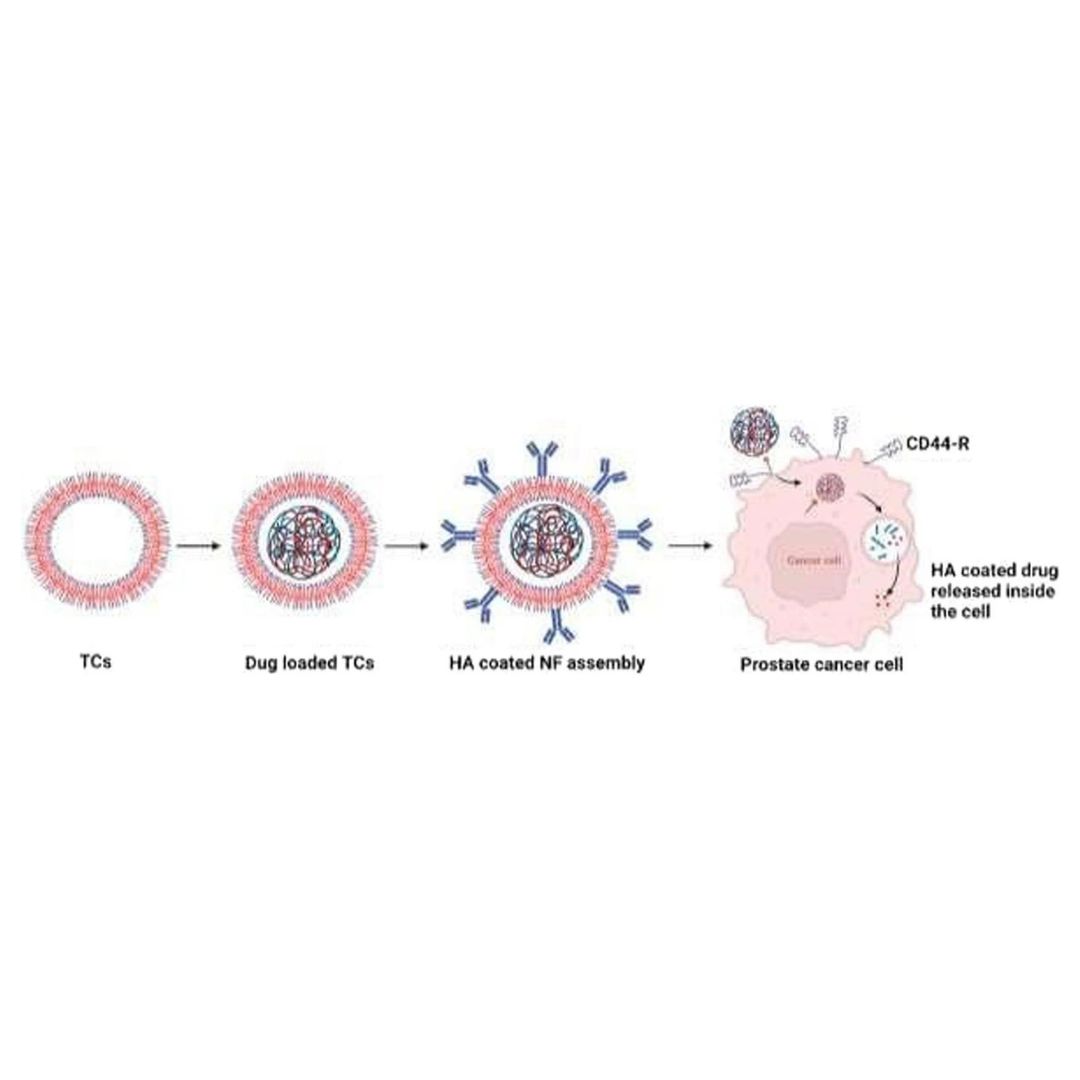
Evaluation of the anticancer potential of CD44 targeted vincristine nanoformulation in prostate cancer xenograft model: a multi-dynamic approach for advanced pharmacokinetic evaluation
The in vivo anticancer potential of vincristine (VC) loaded, thiolated chitosan-based nanoformulation (NFs) with an outer hyaluronic acid (VC-loaded in TCs-HA) coating was studied in prostate cancer (PC) xenograft in the immunosuppressed rat model induced by PC3 cell lines.

The application of AI-driven Drug Discovery technology for molecular optimization of nuclear receptor ligands
Nuclear receptors (NRs) are a superfamily of transcription factors whose activity is regulated upon the binding of a specific ligand.

Development of a Discriminative Dissolution Method, Using In-Silico Tool for Hydrochlorothiazide and Valsartan Tablets
Hydrochlorothiazide (HTZ) and Valsartan (VAL) are poorly soluble drugs in BCS classes IV and II.

June 2023 GastroPlus Newsletter
Workshops and Webinars

Predictive Potential of Acido-Basic Properties, Solubility and Food on Bioequivalence Study Outcome: Analysis of 128 Studies
Risk assessment related to bioequivalence study outcome is critical for effective planning from the early stage of drug product development.

Modeling time‐delayed concentration‐QT effects with ACT ‐1014‐6470, a novel oral complement factor 5a receptor 1 (C5a 1 receptor) antagonist
The novel oral complement factor 5a receptor 1 antagonist ACT- 1014- 6470 was well tolerated in single- and multiple- ascending dose studies, including 24 h Holter electro-cardiogram (ECG) recordings evaluating its cardiodynamics based on data from singledoses of 30– 200 mg and twice- daily (b.i.d.) dosing of 30– 120 mg for 4.5 days.

Gastroplus and HSPiP Oriented Predictive Parameters as the Basis of Valproic Acid Loaded Mucoadhesive Cationic Nanoemulsion Gel for Improved Nose-To-Brain Delivery to Control Convulsion in Human
Oral and parenteral delivery of first-line anticonvulsant Valproic cid (VA) are associated with serious adverse effects, high hepatic metabolism, high clearance, and low bioavailability in brain.

June 2023 News/Events
Explore modeling and simulation summer school opportunities inside
