This manuscript highlights key sessions from the In Vitro Release and Dissolution Testing (IVRDT) and Oral Biopharmaceutics and Absorption Modeling (OBAM) communities at the 2025 AAPS PharmSci 360 Annual Meeting (November 9–12, San Antonio, TX).
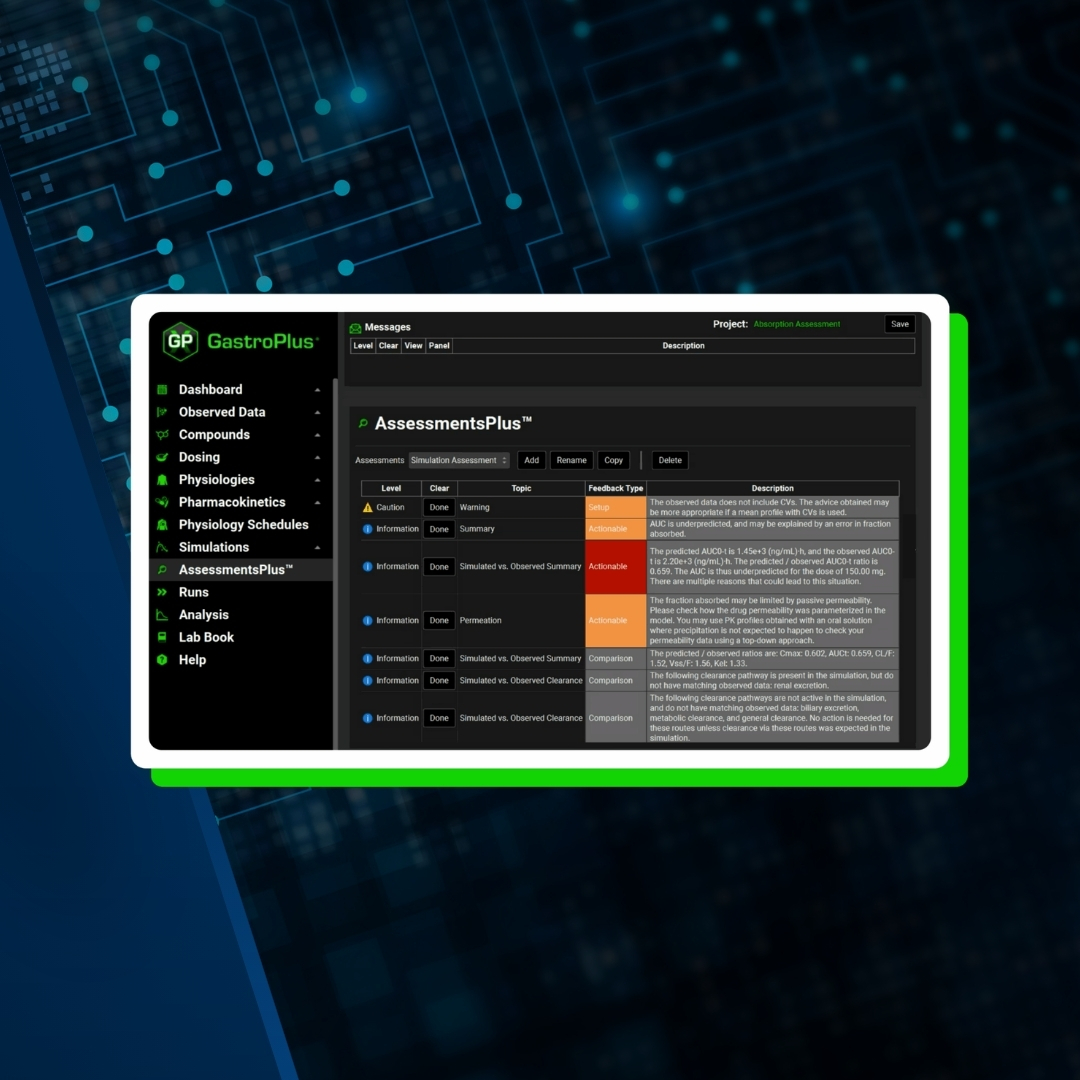
GP AssessmentsPlus™ Flyer
The AssessmentsPlus module in GastroPlus provides structured, expert-guided evaluation of your PBPK and PBBM models.

Project Optimus: Oncology Dose Optimization Based on Clinical Pharmacology Strategy and MIDD Approach
Since the FDA initiated Project Optimus, considerable effort has been stimulated to better select doses of new oncology products.

Why Model Transparency Matters in Regulatory Decision-Making
In high-stakes decision-making, particularly within regulatory submissions to agencies such as the FDA, PMDA, EMA, Health Canada, MHRA, ANVISA, models serve as a weight of evidence approach rather than exploratory tools.

Model-Based Virtual Clinical Trial Reveals Renal Impairment and Body Size as Key Determinants of Pharmacokinetic Variability and Drug-Drug Interaction Risk in Propranolol Therapy
Propranolol (PROP) is a non-selective β-blocker widely prescribed for cardiovascular and neurological disorders.

Impact of Preclinical Verification in Physiologically Based Pharmacokinetic Modelling for First-in-Human Predictions: Application of a Published Model Building Strategy for Five Compounds
Physiologically based pharmacokinetic modelling is routinely used in the pharmaceutical industry and has an impact on drug labels.

Simulations Plus to Participate in the 23rd Annual Craig-Hallum Institutional Investor Conference
Simulations Plus will be participating in the 23rd Annual Craig-Hallum Institutional Investor Conference.
QSP Flex Flyer
Which QSP model option is right for you?

Enrichment, Response Amplification, Separation, and Inclusion: Quantifying Trial Design Trade-offs in SLE with QSP SimPops Modeling
Systemic lupus erythematosus (SLE) trials suffer from equivocal or failed trial outcomes.

Pulmonary Physiological-Based Pharmacokinetic (PBPK) Modeling for Clinical Systemic and Regional PK Prediction of Inhaled Nintedanib Drug Products
Nintedanib (Nint), a tyrosine kinase inhibitor, is approved by the FDA for the treatment of idiopathic pulmonary fibrosis (IPF) by administration as an oral tablet.

Mechanistic PBPK Modeling Identifies Oral Microenvironment Determinants of Buccal Midazolam Exposure
Buccal drug delivery relies on prolonged mucosal contact for absorption, sensitive to interindividual chemical and physical variability within the oral microenvironment (Hua, 2019; Shipp et al., 2022).

GP Metabolism & Transporter Module Flyer
Extend your mechanistic modeling to fully utilize your in vitro data and model complex scenarios.

Simulations Plus and NVIDIA Collaborate to Scale GPU-Accelerated, AI-Assisted Modeling Workflows
Combines validated scientific engines with accelerated computing and AI to enable more scalable, integrated modeling workflows across the drug development lifecycle

Bridging the Gap Between In Vitro and In Vivo: A Mechanistic Approach to Dissolution in PBBM Modeling
For decades, the pharmaceutical industry has pursued a reliable bridge between in vitro dissolution and in vivo performance.

Development of a Physiologically Based Pharmacokinetic (PBPK) Simulation Model for Nicotine
Tobacco is a major cause of chronic diseases such as lung cancer, cardiovascular disease, and chronic obstructive pulmonary disease worldwide.

Advancing Pharmacoequity in Low- and Middle-Income Countries via Model Informed Drug Development
Across low- and middle-income countries (LMICs) inequities in access to safe, effective medicines persist due to limited regulatory capacity, inadequate financing, and insufficient local data.

Physiologically Based Pharmacokinetic Modeling to Predict Human Pharmacokinetics of a Novel Mithramycin Analog for Ewing Sarcoma
To develop and verify a physiologically based pharmacokinetic (PBPK) modeling strategy for mithramycin (MTM) and its analog, MTMSA-Trp, with the aim of projecting first-in-human plasma pharmacokinetics and supporting the translational development of MTMSA-Trp for Ewing sarcoma treatment.

Accelerated Discovery of Novel RORγT Modulators Using an AI-Driven Platform Integrating Generative Chemistry and Mechanistic Pk Simulation
Artificial Neural Network Ensemble models were built in ADMET Modeler using data curated from ChEMBL. (A) Distribution of agonist and inverse agonist activities (in -log space). Classification and Regression models for agonists (B, C) and inverse agonists (D, E). Compounds must pass the classification model prior to prediction in the regression model.

Simulations Plus Announces Collaboration with Lonza and U.S. FDA to Advance Predictive Frameworks for Complex Oral Drug Products
Mechanistic modeling approach and experimental integration designed to improve early risk identification, strengthen regulatory confidence, and expand AI-enabled workflows connecting data to decision-making

Simulations Plus Expands Global Access to Model-Informed Drug Development Training Through Its 2026 Spring School
More than 1,400 participants across 65+ countries underscores accelerating adoption of model-informed workflows and growing demand for applied modeling expertise
