In this video we'll be talking about Injectables Subcutaneous.

GPX™ Tutorial Series: AssessmentsPlus
In this video we'll be talking about AssessmentsPlus.

GPX™ Tutorial Series: P-PSD
In this video we'll be talking about P-PSD.

GPX™ Tutorial Series: Orchestrator
In this video we'll be talking about Orchestrator.

GPX™ Tutorial Series: ACATPlus
In this video we'll be talking about GPX ACATPlus.
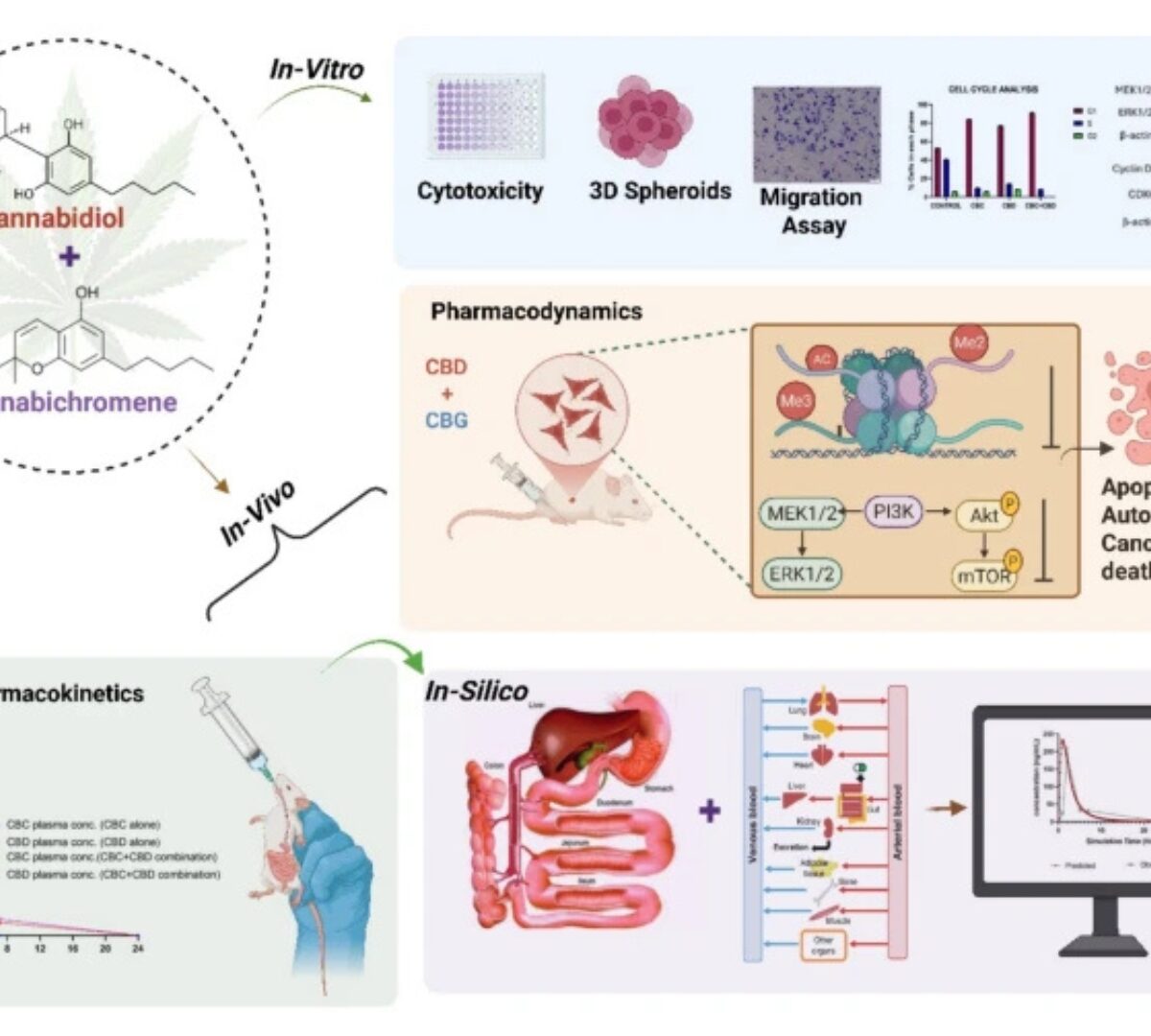
Pharmacokinetic Studies and Synergistic Antitumor Effects of Cannabichromene and Cannabidiol in Drug-Resistant Breast Cancers
Triple-negative breast cancer (TNBC) is highly aggressive with limited treatment options, and resistance to doxorubicin (DOX) further compromises outcomes.

GPX™ Tutorial Series: Introduction to ACAT
In this video we'll be talking about Introduction to ACAT.

GPX™ Tutorial Series: PDPlus Module
In this video we'll be talking about PDPlus Module.

GPX™ Tutorial Series: IVIVCPlus Module
In this video we'll be talking about IVIVCPlus Module.

GPX™ Tutorial Series: PKPlus Module
In this video we'll be talking about PKPlus Module.

GPX™ Tutorial Series: DDI Module
In this video we'll be talking about DDI Module.

GPX™ Tutorial Series: Metabolism and Transport Module
In this video we'll be talking about Metabolism and Transport Module.

GPX™ Tutorial Series: PBPKPlus Module
In this video we'll be talking about PBPKPlus Module.

GPX™ Tutorial Series: Recreating a GPX Project in Legacy GastroPlus
In this video we'll be talking about recreating a GPX project in legacy GastroPlus.

GPX™ Tutorial Series: Lab Book
In this video we'll be talking about lab book.

GPX™ Tutorial Series: Reporting Outputs
In this video we'll be talking about reporting outputs.

GPX™ Tutorial Series: Simplified Absorption and Pharmacokinetics
In this video we'll be talking about simplified absorption and pharmacokinetics.

GPX™ Tutorial Series: Asset Export and Import
In this video we'll be talking about asset export and import.

GPX™ Tutorial Series: Run Modes Pop Simulation and VBE Part 3
In this video we'll be talking about run modes population simulation and VBE.

GPX™ Tutorial Series: Run Modes PSA Part 2
In this video we'll be talking about GPX run modes PSA.
