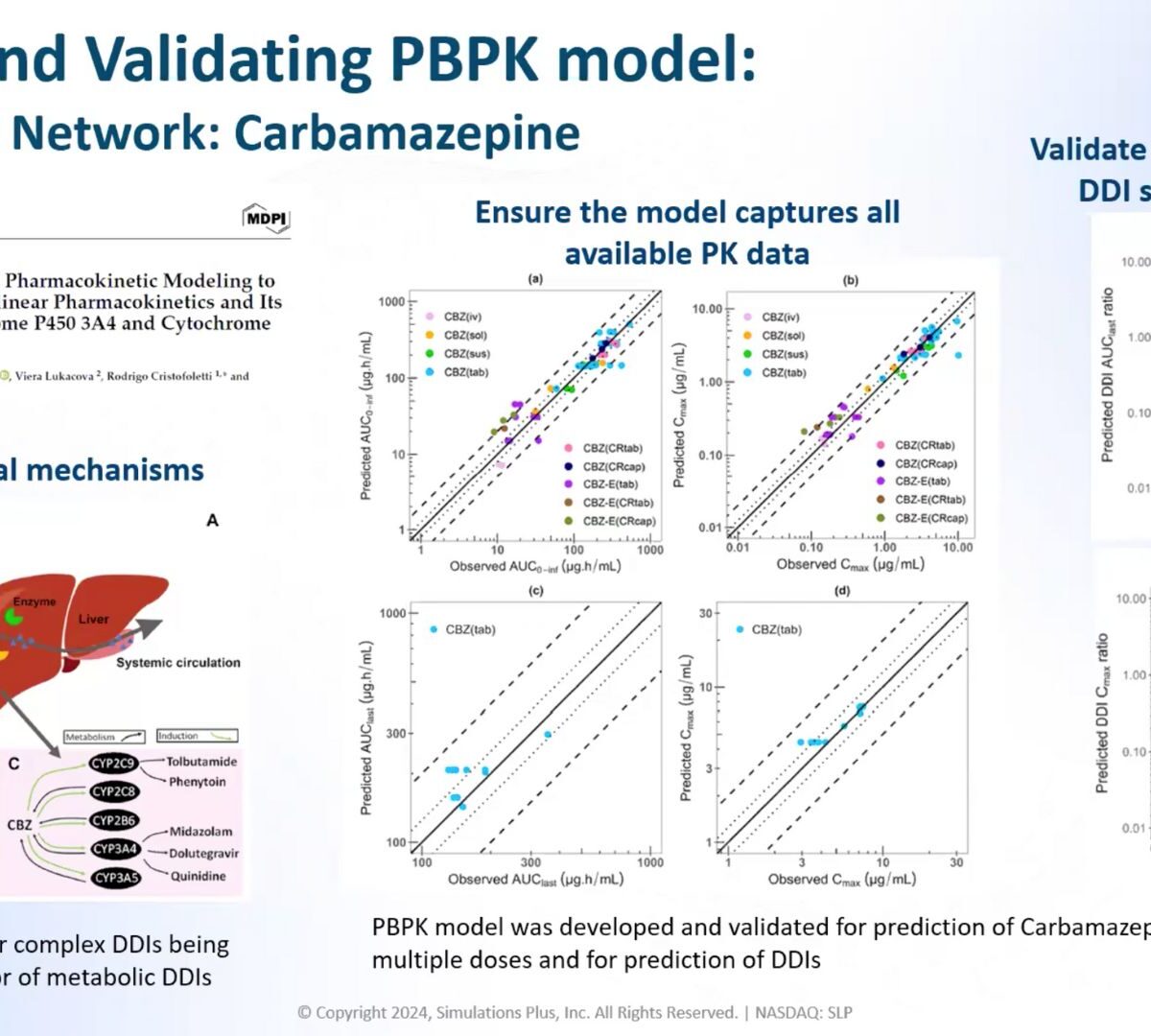
Understanding and predicting drug-drug interactions (DDIs) is crucial for ensuring patient safety and improving drug product development strategies.
DDI Risk Assessment to Inform Your Label Optimize Your Program Timeline & Budget with PBPK Modeling
Every day, scientists in the pharmaceutical industry are tasked with meeting regulatory expectations while also minimizing budget spend by identifying efficiencies for faster development of safer, more effective drugs.
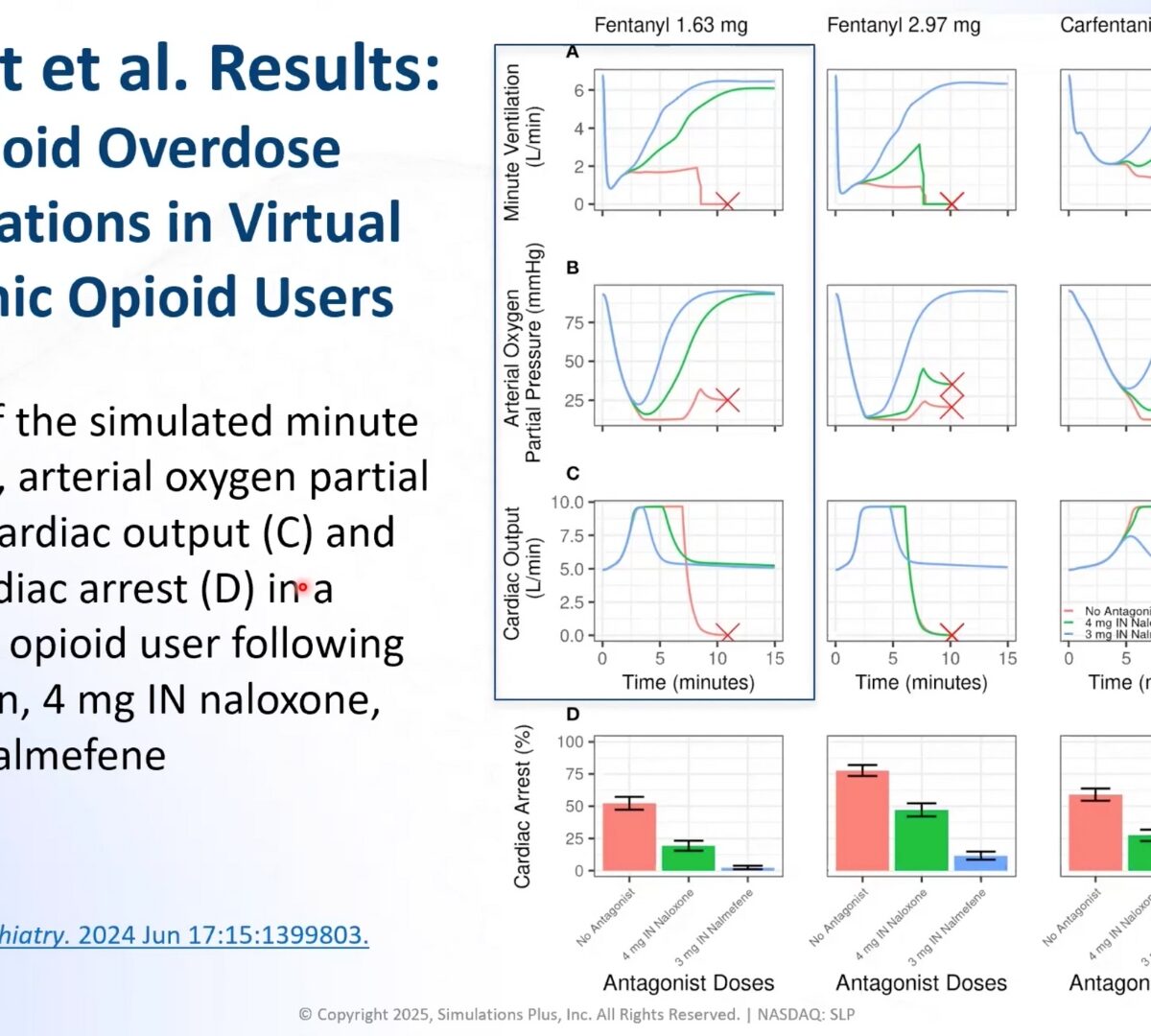
Evaluating the effectiveness of intranasal reversal agents using FDA’s translational model of opioid overdose
The number of opioid overdose deaths in the United States has continued to increase for more than two decades.

De-Risking Clinical Hepatotoxicity in Early Drug Discovery
Hepatotoxicity due to drugs and other xenobiotics, also known as drug-induced liver injury (DILI), is a primary reason for 1) the termination of drug development programs, 2) the delay of approving otherwise efficacious drugs by requiring large and expensive safety-focused clinical trials, 3) the restriction on the clinical use of approved drugs by the inclusion of black box warnings, and 4) the removal of approved drugs from the market.

New Features of the IVIVC Module on the GPX Platform
This webinar, in Portuguese, will explore the IVIVC (in vitro–in vivo correlation) module on the innovative GPX platform.

Ocular Drug Discovery & Development: How Modeling & Simulation is Driving and Optimizing Complex Formulations
The development of ophthalmic drug products is challenging due to the complexity of the ocular system, the lack of sensitive testing to evaluate the interplay of its physiology with ophthalmic drugs, and measurement limitations associated with ocular pharmacokinetics.

Leveraging PBPK/PBBM in Support of BCS Class 3 Biowaivers
It is critical to understand the perspectives and key criteria of global regulatory agencies in the consideration of biowaivers, but many companies have knowledge gaps that hinder their success.

AI for Pharmacological Modeling
Pharmacological modeling plays a key role in drug development by guiding decisions on dosing to optimize safety and efficacy and ultimately streamlining the path to successful therapeutic interventions.

The Application of Artificial Intelligence/Machine Learning in Drug Development and Regulation
Artificial intelligence/machine learning (AI/ML) are increasingly being utilized in drug development.

ADMET Predictor®12: Predict with Confidence
Stay ahead of the competition with ADMET Predictor® 12!

Treating Systemic Lupus Erythematosus: How QSP Can Support Drug Development & Clinical Trial Design
Systemic Lupus Erythematosus (SLE) is a chronic autoimmune disease that affects more than five million people worldwide.

Demystifying GPX™ Installation: Expert Insights and Practical Tips
Get ready to turbocharge your scientific team’s PBBM PBPK modeling technology with the latest version of GastroPlus®!

GPX™ The Next Level PBPK Platform
It’s here—the latest version of GastroPlus®, GPX. And this is your chance to see it in action.

QSP and Oncology: How QSP Modeling Can Accelerate Therapy Development for Solid Tumors
A major obstacle in drug development is the combinatorial complexity of available oncology therapies under development for solid tumors.

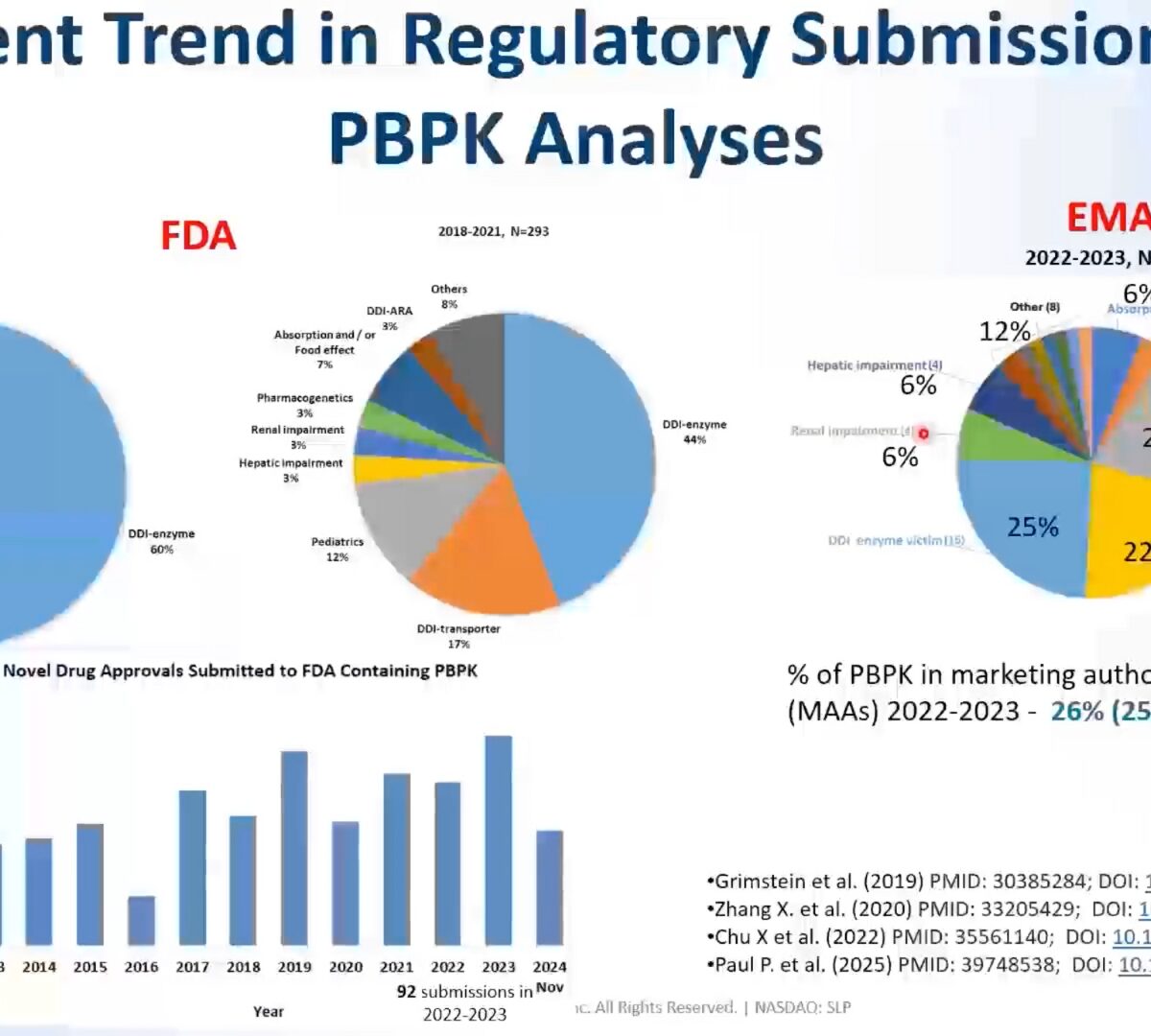
DDIs: How to Use Your Predictions to Support Regulatory Submission
Identifying potential drug-drug interactions (DDIs) is critical to your program's success.

International Perspectives on Model-Informed Precision Dosing: From the Data to the Patients
Identifying the right dose of the right drug for the right patient is a central objective of pharmaceutical drug development.

Unlocking the Power of PBPK Modeling: PBPK for First-in-Human and Beyond
The mechanistic translation of nonclinical pharmacokinetic data to humans can make or break the success of your clinical plan.

Weight Loss Without Nausea: How QSP Modeling Can Optimize Obesity Treatments
Ozempic and Wegovy have been in the news for months as the first highly effective weight loss drugs on the market. However, a noted side effect—nausea—can make it difficult for patients to adhere to proper use.

Using QST Predictions to Optimize Your Drug, De-Risk Your Regulatory Submission, and Guide R&D
After investing years into developing and optimizing a new drug, it’s some of the worst news you can hear: your clinical trials are showing potential liver toxicity.

Mechanistic dissolution data modeling for PBBM introduction: The P-PSD approach
Collect input data for dissolution modeling and test multiple conditions