Since the publication of the ICH E14 guidance in 2015, QT interval prolongation as-sessment can be carried out with a concentration-QTc modeling approach as part of single- or mul-tiple- dose escalation studies, instead of conducting a thorough QT/QTc study.

mlxDesignEval: A novel R package for design evaluation based on MonolixSuite, and its comparison to popED and PFIM
Designing clinical trials to support population PK/PD modeling requires careful choices of sampling times, number of subjects, dose groups and other trial features to
ensure precise parameter estimation - with low relative standard errors [1].

Developing Biosimilars: How to Design PK and PD Similarity Studies Using Modeling & Simulation
Biosimilar development is crucial to making lifesaving treatments more affordable and accessible to patients—and researchers are under increasing pressure to reduce cost and time without compromising scientific rigor

GPX™ Tutorial Series: Introduction to Reprise Licensing
In this video we'll be talking about Introduction to Reprise Licensing.

GPX™ Tutorial Series: Injectables Intramuscular
In this video we'll be talking about Injectables Intramuscular.

GPX™ Tutorial Series: Injectables Subcutaneous
In this video we'll be talking about Injectables Subcutaneous.

GPX™ Tutorial Series: AssessmentsPlus
In this video we'll be talking about AssessmentsPlus.

GPX™ Tutorial Series: P-PSD
In this video we'll be talking about P-PSD.

GPX™ Tutorial Series: Orchestrator
In this video we'll be talking about Orchestrator.

GPX™ Tutorial Series: ACATPlus
In this video we'll be talking about GPX ACATPlus.
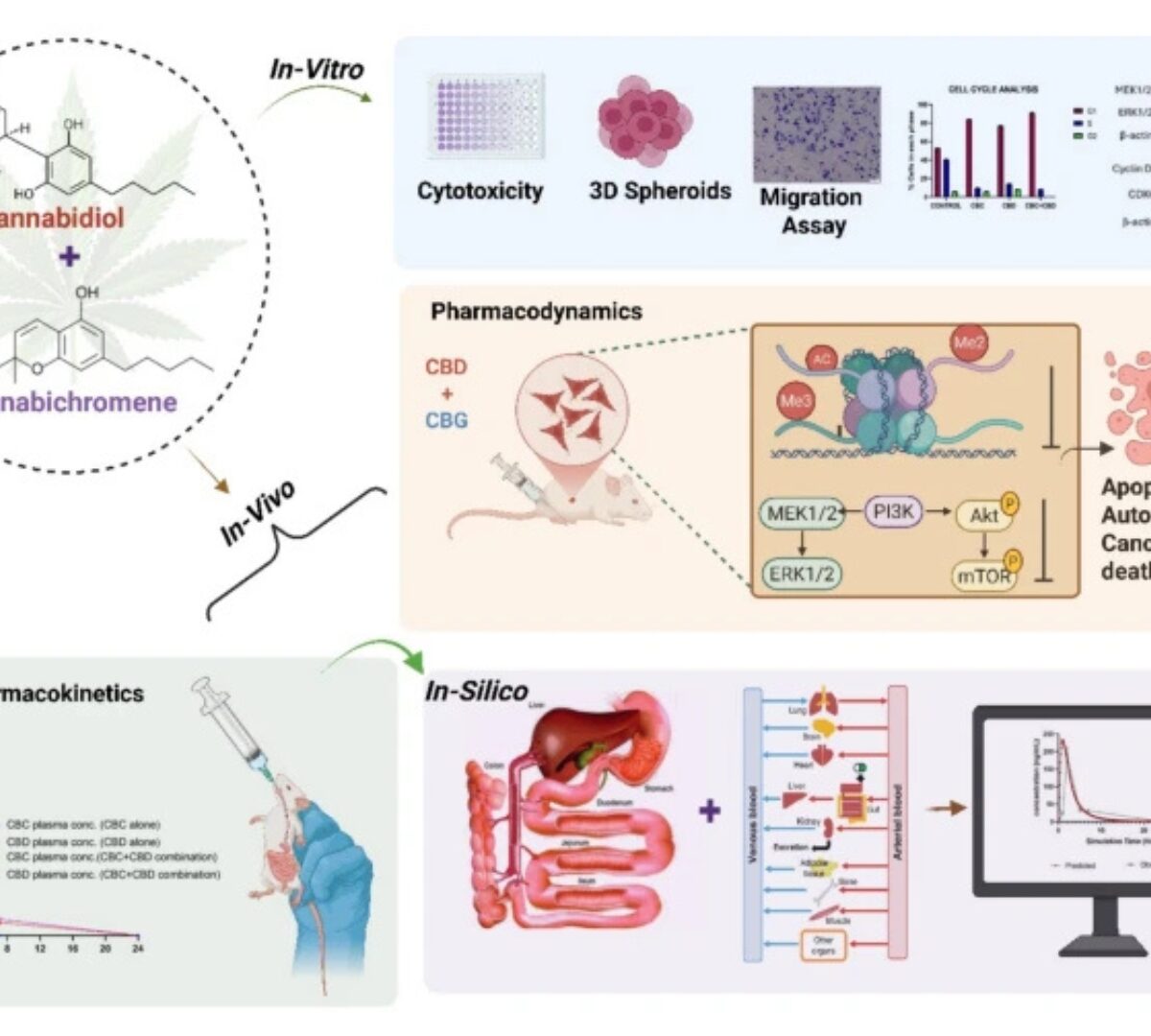
Pharmacokinetic Studies and Synergistic Antitumor Effects of Cannabichromene and Cannabidiol in Drug-Resistant Breast Cancers
Triple-negative breast cancer (TNBC) is highly aggressive with limited treatment options, and resistance to doxorubicin (DOX) further compromises outcomes.

GPX™ Tutorial Series: Introduction to ACAT
In this video we'll be talking about Introduction to ACAT.

GPX™ Tutorial Series: PDPlus Module
In this video we'll be talking about PDPlus Module.

GPX™ Tutorial Series: IVIVCPlus Module
In this video we'll be talking about IVIVCPlus Module.

GPX™ Tutorial Series: PKPlus Module
In this video we'll be talking about PKPlus Module.

GPX™ Tutorial Series: DDI Module
In this video we'll be talking about DDI Module.

GPX™ Tutorial Series: Metabolism and Transport Module
In this video we'll be talking about Metabolism and Transport Module.

GPX™ Tutorial Series: PBPKPlus Module
In this video we'll be talking about PBPKPlus Module.

GPX™ Tutorial Series: Recreating a GPX Project in Legacy GastroPlus
In this video we'll be talking about recreating a GPX project in legacy GastroPlus.

GPX™ Tutorial Series: Lab Book
In this video we'll be talking about lab book.
