Probenecid, an oral uricosuric agent, is being developed as a broad-spectrum antiviral and was evaluated for potential suppressive effects of SARS-Cov-2 replication in a Phase 2 study in patients with symptomatic, mild-to-moderate COVID-19

Comparison of Population Pharmacokinetic Platforms, Monolix and Phoenix for Cephalexin in Infants
Cephalexin is a commonly used antibiotic for pediatric populations, however, data to guide effective oral dosing in young infants are limited.
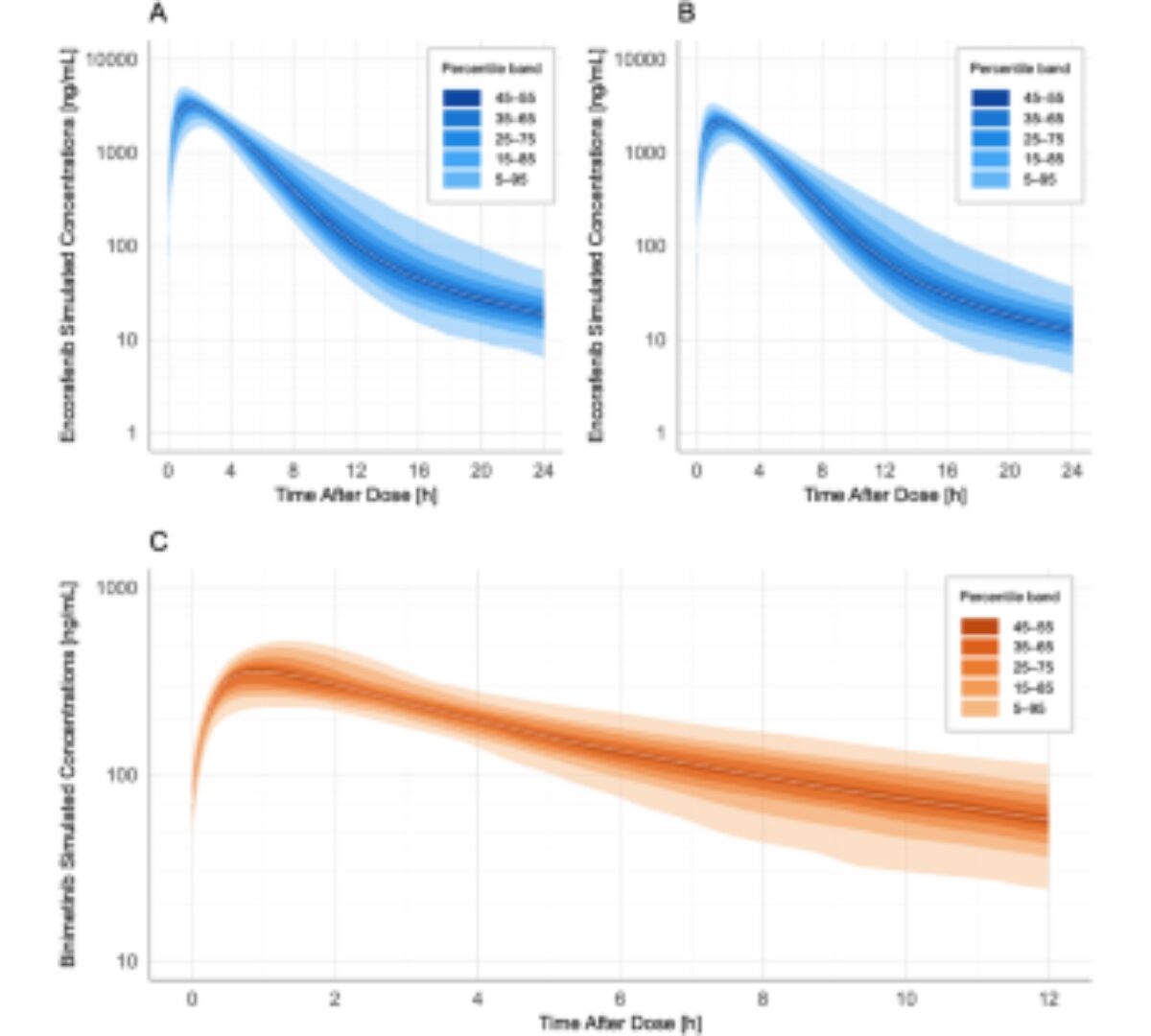
Population Pharmacokinetics of Encorafenib and Binimetinib in Real-World Patients with BRAFV600E/K-Mutant Metastatic Melanoma
Encorafenib and binimetinib pharmacokinetic (PK) studies in real-world cancer patients remain scarce.
Integrating Systemic Toxicity and Toxicokinetic Data to Inform the Need for Subchronic Dog Studies in Human Health Safety Assessments of Agrochemicals
Regulatory testing for agrochemicals has traditionally included a 90-day toxicity study in a non-rodent species, usually the dog.

ADMET Predictor® Tutorial Series: HTPK Part 1
In this first part of the HTPK series, we demonstrate how to run high-throughput pharmacokinetic (HTPK) simulations directly from chemical structures. Learn to calculate fraction absorbed (%FA), fraction bioavailable (%FB), and key PK parameters like Cmax, Tmax, and clearance across multiple doses.

ADMET Predictor® Tutorial Series: MedChem Studio™ Part 2
In the second part of the MedChem Studio™ series, we demonstrate how to evaluate compound subsets and optimize virtual libraries. This tutorial covers distribution analysis to compare virtual vs. exemplified compounds and introduces matched molecular pair (MMP) analysis for property cliff detection.

ADMET Predictor® Tutorial Series: MedChem Studio™ Part 1
In this first installment of the MedChem Studio™ series, we demonstrate powerful clustering and group analysis tools. Learn how to organize large data sets into chemotypes, align scaffolds for visual comparison, and perform R-group decomposition to identify SAR trends

ADMET Predictor® Tutorial Series: Predicting Metabolites
In this video, we explore advanced tools for predicting drug metabolites across major enzyme classes, including Cytochrome P450 (SIP), Aldehyde Oxidase (AOX), and UGTs. You will learn how to identify atomic sites of metabolism, interpret scoring systems, and visualize structural transformations within MedChem Designer™.

ADMET Predictor® Tutorial Series: Model Editor
In this video, we provide a detailed overview of the Model Editor, explaining how to manage prediction modules and interpret applicability domains. Key topics include descriptor sensitivity analysis (DSA), structure sensitivity analysis (SSA), and regression uncertainty.

ADMET Predictor® Tutorial Series: Predicting pKa
In this video, Simulations Plus demonstrates how to predict pKa values and their critical impact on pharmacokinetic properties like solubility and permeability. You will learn how to interpret microstate displays, analyze ionization curves, and understand how pKa influences drug behavior in the GI tract.

ADMET Predictor® Tutorial Series: Working with Data and Graphs
In this video, Simulations Plus explains how to examine data and utilize advanced graphing tools within the platform. Key features include managing spreadsheet columns, creating custom tabs, and visualizing molecular properties through various chart types.

ADMET Predictor® Tutorial Series: Calculating Properties
In this video, we explore the core functionality of calculating chemical and biological properties. It covers selecting specific models, adjusting pH settings, and configuring multi-threading to maximize processing speed.

ADMET Predictor® Tutorial Series: MedChem Designer
In this video, we provide an interactive tutorial on using MedChem Designer to edit, create, and save chemical structures. You will also learn how to calculate ADMET properties directly within the designer and import structures from online resources like DrugBank.

ADMET Predictor® Tutorial Series: Working with Files
In this video, we demonstrate how to open chemical structure files and import external property data. You will also see how to save progress using proprietary XDK session files.

Integrating Diverse Clinical Data into a Single Virtual Population with Thales™
As the complexity of drug development increases, researchers need quantitative systems pharmacology (QSP) models that incorporate virtual populations and can capture data spanning numerous clinical trial
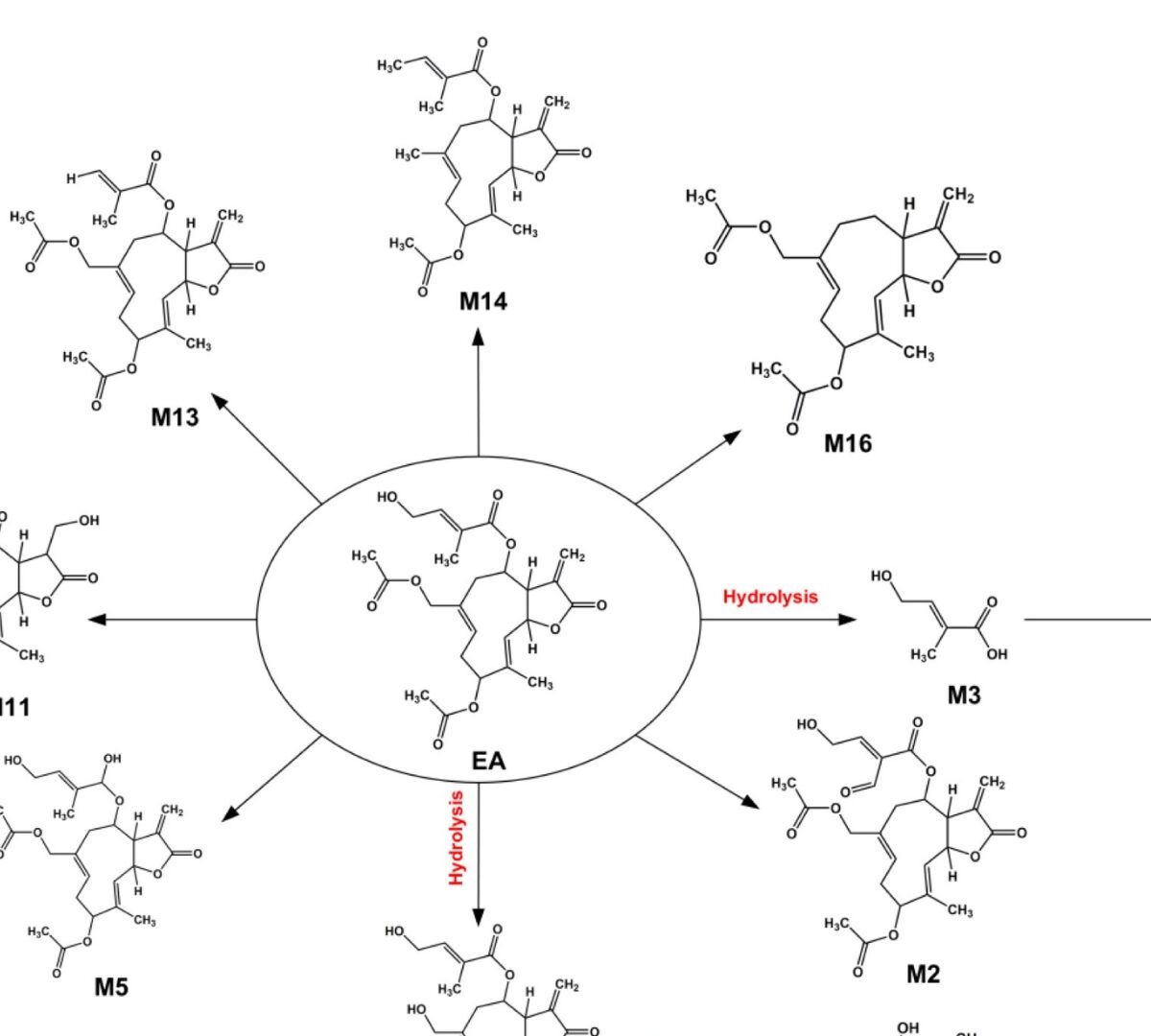
Metabolic Profiling and Detoxification of Eupalinolide A and B in Human Liver Microsomal Systems
Eupalinolide A (EA, Z-configuration) and Eupalinolide B (EB, E-configuration) are cis-trans isomeric sesquiterpenoid monomers isolated from Eupatorium lindleyanum DC. (Asteraceae).

Simulations Plus Convenes Industry and Regulatory Leaders to Define Responsible AI in MIDD
Expert panel at the 2026 ASCPT Annual Meeting brings together leaders with industry and regulatory experience to explore practical, accountable AI implementation

Development of a Quantitative Systems Toxicology Model to Predict Drug-Induced Liver Injury in Pediatrics
Drug-induced liver injury (DILI) is an underrecognized cause of pediatric liver disease which accounts for almost 20% of pediatric acute liver failure cases, and is a major reason for liver transplantation in the USA [1].

Prospective Physiologically Based Pharmacokinetic Modeling Predictions of First-in-Patient Study PK: Examples and Application
The examples shown here are intended to provide an update on the accuracy of the method for prospective predictions.

A Pediatric Pbpk Model of Atropine Gel To Predict Atropine Levels in Children With Neurological Disorders After Administration to Oral Cavity
Sialorrhea, or excessive salivation, is a chronic and serious problem in children with cerebral palsy (CP) and neurodevelopmental disorders.[1–5] Sialorrhea occurs in up to 60% of children with CP...
